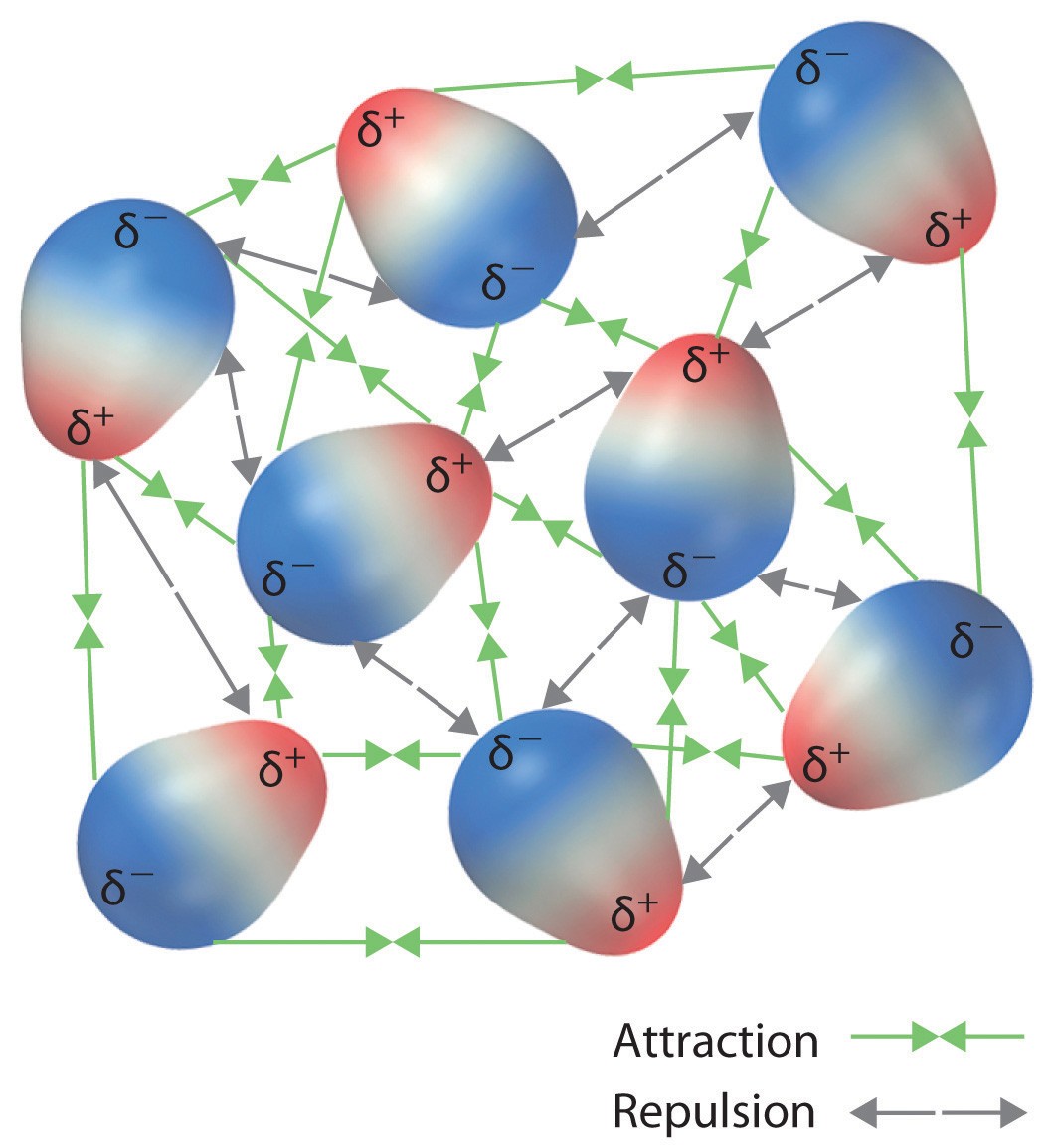
Which Statement Compares The Attractive Forces That Hold Particles Together? Understanding these forces is crucial in grasping the properties of matter. At COMPARE.EDU.VN, we provide detailed comparisons to help you differentiate between various attractive forces, including dipole-dipole interactions, London dispersion forces, and hydrogen bonds. Exploring these fundamental forces enhances understanding, application, and insightful analysis.
1. Introduction to Attractive Forces
Attractive forces, also known as intermolecular forces, play a vital role in determining the physical properties of substances. These forces exist between molecules and are responsible for holding them together in liquid and solid states. Unlike intramolecular forces, such as covalent bonds that hold atoms together within a molecule, intermolecular forces are weaker but essential for understanding material behavior. These forces determine the boiling points, melting points, viscosity, and surface tension of different substances.
1.1 Importance of Understanding Intermolecular Forces
Understanding intermolecular forces is critical for several reasons:
- Predicting Physical Properties: Intermolecular forces help predict whether a substance will be a solid, liquid, or gas at a given temperature.
- Explaining Material Behavior: They explain why some substances have high boiling points while others have low boiling points.
- Designing New Materials: Understanding these forces is essential for designing new materials with specific properties.
- Biological Processes: They play a crucial role in biological systems, such as protein folding and DNA structure.
1.2 Overview of Different Types of Attractive Forces
There are primarily three types of attractive forces that hold particles together:
- Dipole-Dipole Interactions: Occur between polar molecules.
- London Dispersion Forces: Exist between all molecules, including nonpolar molecules.
- Hydrogen Bonds: A special type of dipole-dipole interaction that occurs when hydrogen is bonded to highly electronegative atoms.
Understanding the distinctions and strengths of these forces is essential for predicting and explaining the properties of various substances.
2. Dipole-Dipole Interactions
Dipole-dipole interactions occur between polar molecules, which have a positive end and a negative end due to unequal sharing of electrons. These interactions arise when the positive end of one molecule attracts the negative end of another.
2.1 Definition and Explanation
A dipole moment is created when there is a separation of charge within a molecule. This occurs when atoms in a molecule have different electronegativities, leading to unequal sharing of electrons. The more electronegative atom attracts electrons more strongly, resulting in a partial negative charge (δ-) on that atom and a partial positive charge (δ+) on the less electronegative atom. Molecules with dipole moments are called polar molecules.
When polar molecules are close together, they tend to align themselves so that the positive end of one molecule is near the negative end of another. This alignment results in an attractive force between the molecules, known as a dipole-dipole interaction.
2.2 Factors Affecting the Strength of Dipole-Dipole Interactions
The strength of dipole-dipole interactions depends on several factors:
- Magnitude of Dipole Moment: The larger the dipole moment, the stronger the interaction. Molecules with highly polar bonds will have stronger dipole-dipole interactions.
- Distance Between Molecules: The closer the molecules, the stronger the interaction. Dipole-dipole interactions decrease rapidly with increasing distance.
- Molecular Orientation: The alignment of molecules affects the strength of the interaction. Optimal alignment occurs when the positive and negative ends of adjacent molecules are directly aligned.
2.3 Examples of Substances with Dipole-Dipole Interactions
Several common substances exhibit dipole-dipole interactions:
- Hydrogen Chloride (HCl): A polar molecule with a significant dipole moment due to the electronegativity difference between hydrogen and chlorine.
- Acetone (CH3COCH3): A polar molecule with a carbonyl group (C=O) that creates a dipole moment.
- Acetonitrile (CH3CN): A polar molecule with a cyano group (C≡N) that generates a substantial dipole moment.
These substances have higher boiling points compared to nonpolar substances of similar molecular weight due to the presence of dipole-dipole interactions.
3. London Dispersion Forces
London dispersion forces, also known as van der Waals forces, are temporary attractive forces that exist between all molecules, whether polar or nonpolar. These forces arise from instantaneous fluctuations in electron distribution.
3.1 Definition and Explanation
Even in nonpolar molecules, the electron distribution is not always perfectly uniform. At any given instant, the electrons may be unevenly distributed, creating a temporary, instantaneous dipole moment. This instantaneous dipole can induce a dipole in a neighboring molecule, leading to an attractive force between the two molecules. These temporary dipoles are constantly forming and disappearing, resulting in weak but significant attractive forces.
3.2 Factors Affecting the Strength of London Dispersion Forces
The strength of London dispersion forces depends on several factors:
- Molecular Size and Surface Area: Larger molecules with greater surface areas have more electrons and more opportunities for temporary dipoles to form. As a result, larger molecules generally have stronger London dispersion forces.
- Molecular Shape: Molecules with more extended shapes can come into closer contact with neighboring molecules, leading to stronger London dispersion forces. Compact, spherical molecules have smaller surface areas and weaker interactions.
- Polarizability: Polarizability refers to the ease with which the electron distribution in an atom or molecule can be distorted. Larger atoms and molecules with more loosely held electrons are more polarizable and exhibit stronger London dispersion forces.
3.3 Examples of Substances with London Dispersion Forces
Examples of substances where London dispersion forces are significant:
- Noble Gases (He, Ne, Ar, Kr, Xe): Noble gases are nonpolar and held together only by London dispersion forces. Their boiling points increase with increasing atomic size due to greater polarizability.
- Alkanes (CH4, C2H6, C3H8, etc.): Alkanes are nonpolar hydrocarbons with increasing boiling points as the carbon chain length increases, reflecting stronger London dispersion forces.
- Nonpolar Halogens (F2, Cl2, Br2, I2): These diatomic molecules exhibit London dispersion forces, with boiling points increasing from fluorine to iodine due to increasing molecular size and polarizability.
The boiling points of these substances are generally lower compared to substances with dipole-dipole interactions or hydrogen bonds, but London dispersion forces are always present and contribute to the overall attractive forces.
4. Hydrogen Bonds
Hydrogen bonds are a special type of dipole-dipole interaction that occurs when a hydrogen atom is bonded to a highly electronegative atom, such as oxygen (O), nitrogen (N), or fluorine (F). This creates a strong dipole moment, with the hydrogen atom carrying a significant partial positive charge (δ+).
4.1 Definition and Explanation
In a hydrogen bond, the partially positive hydrogen atom is attracted to a lone pair of electrons on another electronegative atom (O, N, or F) in a neighboring molecule. This attraction is stronger than typical dipole-dipole interactions due to the high polarity of the bond and the small size of the hydrogen atom, allowing for close proximity between the molecules.
4.2 Factors Affecting the Strength of Hydrogen Bonds
The strength of hydrogen bonds depends on several factors:
- Electronegativity of the Atom: The greater the electronegativity of the atom bonded to hydrogen, the stronger the hydrogen bond. Fluorine forms the strongest hydrogen bonds, followed by oxygen and nitrogen.
- Lone Pair Availability: The presence of available lone pairs on the acceptor atom is essential for forming hydrogen bonds.
- Molecular Geometry: The geometry of the molecules must allow for proper alignment between the hydrogen bond donor and acceptor.
- Distance and Angle: Shorter distances and more linear arrangements result in stronger hydrogen bonds.
4.3 Examples of Substances with Hydrogen Bonds
Substances that exhibit hydrogen bonding include:
- Water (H2O): Water is a prime example of hydrogen bonding. Each water molecule can form up to four hydrogen bonds with neighboring molecules, leading to its unique properties such as high boiling point, surface tension, and density anomalies.
- Ammonia (NH3): Ammonia molecules can form hydrogen bonds with each other, although not as strong as in water.
- Alcohols (ROH): Alcohols, such as ethanol and methanol, contain a hydroxyl group (OH) that can participate in hydrogen bonding.
- Proteins and DNA: Hydrogen bonds play a crucial role in stabilizing the structures of proteins and DNA, influencing their biological functions.
Hydrogen bonds significantly elevate the boiling points and melting points of substances compared to similar molecules without hydrogen bonding.
5. Comparing the Different Types of Attractive Forces
To understand which statement compares the attractive forces that hold particles together, it is essential to compare their relative strengths and characteristics.
5.1 Relative Strengths of Attractive Forces
The relative strengths of the different types of attractive forces are as follows, from strongest to weakest:
- Hydrogen Bonds: Typically the strongest type of intermolecular force, with energies ranging from 15 to 25 kJ/mol.
- Dipole-Dipole Interactions: Weaker than hydrogen bonds but stronger than London dispersion forces, with energies typically ranging from 2 to 10 kJ/mol.
- London Dispersion Forces: Generally the weakest type of intermolecular force, with energies ranging from 0.1 to 10 kJ/mol, depending on molecular size and shape.
It’s important to note that these values are approximate and can vary depending on the specific molecules and conditions.
5.2 Factors Influencing the Predominant Attractive Force
Several factors determine which type of attractive force will be predominant in a given substance:
- Molecular Polarity: Polar molecules exhibit dipole-dipole interactions, while nonpolar molecules rely on London dispersion forces. Hydrogen bonding occurs when hydrogen is bonded to highly electronegative atoms.
- Molecular Size and Shape: Larger molecules with greater surface areas tend to have stronger London dispersion forces.
- Presence of Hydrogen Bond Donors and Acceptors: Molecules with hydrogen atoms bonded to O, N, or F can form hydrogen bonds.
5.3 Examples Comparing Different Substances
Consider the following examples to illustrate the differences in attractive forces:
- Methane (CH4) vs. Water (H2O): Methane is nonpolar and exhibits only London dispersion forces, resulting in a low boiling point (-161.5°C). Water, on the other hand, can form hydrogen bonds, leading to a much higher boiling point (100°C).
- Ethanol (C2H5OH) vs. Dimethyl Ether (CH3OCH3): Ethanol can form hydrogen bonds due to the presence of a hydroxyl group (OH), while dimethyl ether cannot. As a result, ethanol has a higher boiling point (78.3°C) compared to dimethyl ether (-24°C).
- Butane (C4H10) vs. Pentane (C5H12): Both butane and pentane are nonpolar and exhibit London dispersion forces. However, pentane has a larger molecular size and surface area, leading to stronger London dispersion forces and a higher boiling point (36.1°C) compared to butane (-0.5°C).
These examples highlight how the type and strength of attractive forces significantly influence the physical properties of substances.
6. Applications of Understanding Attractive Forces
Understanding attractive forces has numerous practical applications in various fields:
6.1 Material Science
In material science, understanding attractive forces is crucial for designing materials with specific properties:
- Polymers: The properties of polymers, such as strength, flexibility, and melting point, are determined by intermolecular forces between polymer chains.
- Adhesives: Adhesives rely on intermolecular forces to bond materials together. Strong adhesives utilize strong attractive forces to create durable bonds.
- Nanomaterials: Intermolecular forces play a significant role in the self-assembly and stability of nanomaterials.
6.2 Biology and Biochemistry
Attractive forces are essential for many biological processes:
- Protein Folding: Hydrogen bonds, hydrophobic interactions, and other attractive forces determine the three-dimensional structure of proteins, which is critical for their function.
- DNA Structure: Hydrogen bonds between complementary base pairs (adenine-thymine and guanine-cytosine) stabilize the double helix structure of DNA.
- Enzyme-Substrate Interactions: Intermolecular forces facilitate the binding of enzymes to their substrates, enabling biochemical reactions.
6.3 Chemistry and Chemical Engineering
Attractive forces are important in chemical processes and engineering applications:
- Solubility: The solubility of a substance depends on the attractive forces between the solute and solvent molecules.
- Distillation: Distillation processes rely on differences in boiling points, which are determined by intermolecular forces.
- Phase Transitions: Understanding attractive forces is essential for predicting and controlling phase transitions (e.g., melting, boiling, sublimation).
7. Challenges in Comparing Attractive Forces
Despite the importance of understanding attractive forces, there are several challenges in accurately comparing and predicting their effects:
7.1 Complexity of Molecular Interactions
Real-world systems often involve a combination of different types of attractive forces, making it challenging to isolate and quantify the contribution of each force. Molecular interactions can be complex and influenced by multiple factors, such as temperature, pressure, and the presence of other molecules.
7.2 Computational Limitations
Accurately calculating intermolecular forces requires sophisticated computational methods, such as molecular dynamics simulations and quantum mechanical calculations. These methods can be computationally intensive and may not be feasible for large or complex systems.
7.3 Experimental Difficulties
Measuring intermolecular forces directly is difficult due to their small magnitude and short range. Experimental techniques, such as atomic force microscopy and surface force apparatus, can provide valuable information, but they are often limited in their applicability and precision.
8. Innovations and Future Directions
Ongoing research and innovations are addressing the challenges in comparing and understanding attractive forces:
8.1 Advanced Computational Methods
Researchers are developing more efficient and accurate computational methods for calculating intermolecular forces. These methods incorporate advanced algorithms and parallel computing techniques to handle large and complex systems.
8.2 Machine Learning and Data Analysis
Machine learning techniques are being used to analyze large datasets of molecular properties and predict intermolecular forces based on structural and chemical features. This approach can accelerate the discovery of new materials and the optimization of chemical processes.
8.3 Enhanced Experimental Techniques
New experimental techniques are being developed to measure intermolecular forces with higher precision and resolution. These techniques include improved atomic force microscopy, surface plasmon resonance, and other advanced spectroscopic methods.
9. COMPARE.EDU.VN: Your Resource for Detailed Comparisons
Navigating the complexities of attractive forces requires access to accurate and comprehensive information. COMPARE.EDU.VN is dedicated to providing detailed and objective comparisons of scientific concepts, including intermolecular forces. Our resources are designed to help students, professionals, and anyone interested in making informed decisions based on reliable data.
9.1 How COMPARE.EDU.VN Can Help
COMPARE.EDU.VN offers:
- Detailed Comparison Articles: In-depth analyses of different attractive forces, including dipole-dipole interactions, London dispersion forces, and hydrogen bonds.
- Objective Evaluations: Unbiased assessments based on scientific evidence and expert analysis.
- User Reviews and Ratings: Insights from users who have explored these concepts, providing valuable perspectives and real-world applications.
- Comprehensive Information: Access to a wide range of resources, including articles, guides, and tools for comparing different types of attractive forces.
9.2 Benefits of Using COMPARE.EDU.VN
By using COMPARE.EDU.VN, you can:
- Save Time and Effort: Quickly find the information you need without spending hours searching through multiple sources.
- Make Informed Decisions: Access detailed comparisons and objective evaluations to make confident choices.
- Enhance Your Knowledge: Gain a deeper understanding of attractive forces and their applications.
- Stay Up-to-Date: Keep abreast of the latest research and innovations in the field.
10. Conclusion
Understanding the different types of attractive forces that hold particles together is essential for explaining and predicting the properties of matter. Dipole-dipole interactions, London dispersion forces, and hydrogen bonds each play a unique role in determining the physical and chemical behavior of substances. By comparing these forces and understanding their relative strengths and characteristics, we can gain valuable insights into a wide range of phenomena, from material design to biological processes.
10.1 Summary of Key Points
- Dipole-Dipole Interactions: Occur between polar molecules and depend on the magnitude of the dipole moment and the distance between molecules.
- London Dispersion Forces: Exist between all molecules and depend on molecular size, shape, and polarizability.
- Hydrogen Bonds: A special type of dipole-dipole interaction that occurs when hydrogen is bonded to highly electronegative atoms.
- Relative Strengths: Hydrogen bonds > Dipole-dipole interactions > London dispersion forces.
10.2 Final Thoughts
COMPARE.EDU.VN is your trusted resource for comprehensive and objective comparisons of attractive forces. Whether you are a student, researcher, or professional, our platform provides the information and tools you need to make informed decisions and enhance your understanding of the world around you.
For further information or assistance, please contact us:
Address: 333 Comparison Plaza, Choice City, CA 90210, United States
WhatsApp: +1 (626) 555-9090
Website: COMPARE.EDU.VN
Explore compare.edu.vn today and discover the power of informed comparison.
FAQ: Attractive Forces
1. What are attractive forces, and why are they important?
Attractive forces, also known as intermolecular forces, are the forces that hold molecules together in liquids and solids. They are essential for determining the physical properties of substances, such as boiling points, melting points, viscosity, and surface tension.
2. What are the main types of attractive forces?
The main types of attractive forces are:
- Dipole-dipole interactions
- London dispersion forces
- Hydrogen bonds
3. What are dipole-dipole interactions?
Dipole-dipole interactions occur between polar molecules, which have a positive end and a negative end due to unequal sharing of electrons. These interactions arise when the positive end of one molecule attracts the negative end of another.
4. What are London dispersion forces?
London dispersion forces are temporary attractive forces that exist between all molecules, whether polar or nonpolar. These forces arise from instantaneous fluctuations in electron distribution, creating temporary dipoles that induce dipoles in neighboring molecules.
5. What are hydrogen bonds?
Hydrogen bonds are a special type of dipole-dipole interaction that occurs when a hydrogen atom is bonded to a highly electronegative atom, such as oxygen (O), nitrogen (N), or fluorine (F). The partially positive hydrogen atom is attracted to a lone pair of electrons on another electronegative atom in a neighboring molecule.
6. How do hydrogen bonds differ from other types of dipole-dipole interactions?
Hydrogen bonds are stronger than typical dipole-dipole interactions due to the high polarity of the bond and the small size of the hydrogen atom, allowing for close proximity between the molecules.
7. What factors affect the strength of dipole-dipole interactions?
The strength of dipole-dipole interactions depends on:
- Magnitude of the dipole moment
- Distance between molecules
- Molecular orientation
8. What factors affect the strength of London dispersion forces?
The strength of London dispersion forces depends on:
- Molecular size and surface area
- Molecular shape
- Polarizability
9. What factors affect the strength of hydrogen bonds?
The strength of hydrogen bonds depends on:
- Electronegativity of the atom bonded to hydrogen
- Lone pair availability
- Molecular geometry
- Distance and angle
10. Can a molecule have multiple types of attractive forces?
Yes, most molecules experience a combination of attractive forces. For example, a polar molecule may exhibit both dipole-dipole interactions and London dispersion forces. Hydrogen bonding can occur in addition to these forces if the molecule contains hydrogen atoms bonded to highly electronegative atoms (O, N, or F).Understanding these FAQs can further clarify the nuances of attractive forces and their significance in various scientific disciplines.