The Isoelectric Point Compared To Ph is a critical concept in biochemistry, influencing the behavior of amino acids and proteins in various solutions. This article will explore the isoelectric point, its relationship to pH, and its significance in biological systems, inviting you to delve into more comprehensive comparisons at COMPARE.EDU.VN. Understand the pivotal roles of zwitterions, amphoteric properties, and acid-base chemistry.
1. Understanding Isoelectric Point (pI): A Comprehensive Overview
The isoelectric point (pI) is the specific pH at which a molecule carries no net electrical charge. This condition arises because the molecule contains both positive and negative charges, which balance each other out perfectly at this particular pH level. This phenomenon is crucial in understanding how amino acids, proteins, and other amphoteric molecules behave in solutions with varying acidity or alkalinity.
1.1. What is the Isoelectric Point?
The isoelectric point, often denoted as pI, is the pH value at which a molecule, such as an amino acid or protein, carries no net electrical charge. At this specific pH, the total positive charges equal the total negative charges on the molecule.
- Definition: The pH at which a molecule has a net charge of zero.
- Importance: Affects solubility, electrophoretic mobility, and protein interactions.
- Determination: Calculated based on the pKa values of ionizable groups within the molecule.
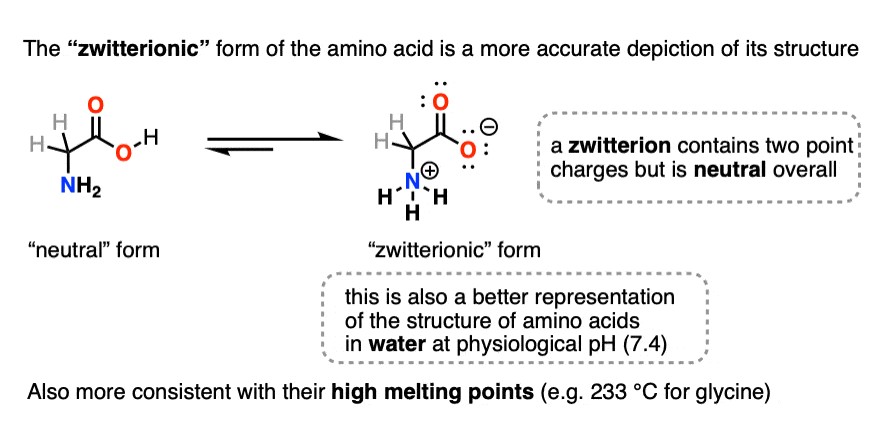
1.2. Zwitterions: The Key to Understanding pI
Amino acids exist predominantly as zwitterions in aqueous solutions. A zwitterion is a molecule that contains both positive and negative electrical charges, even though the overall charge of the molecule is neutral. This amphoteric nature is central to the concept of the isoelectric point.
- Definition: A molecule with both positive and negative charges.
- Formation: Results from the internal transfer of a proton from the carboxyl group to the amino group of an amino acid.
- Significance: Allows amino acids and proteins to act as both acids and bases, influencing their behavior at different pH levels.
1.3. Amphoteric Nature of Amino Acids
Amino acids are amphoteric, meaning they can act as both acids and bases. This dual functionality is due to the presence of both an amino group (NH2) and a carboxyl group (COOH) in their structure. The amino group can accept a proton (acting as a base), while the carboxyl group can donate a proton (acting as an acid).
- Definition: Capable of acting as both an acid and a base.
- Mechanism: Amino group can accept protons, and the carboxyl group can donate protons.
- Impact: Influences their behavior in different pH environments and their buffering capacity.
1.4. Isoelectric Point vs. Isoionic Point
While often used interchangeably, the terms “isoelectric point” and “isoionic point” have subtle differences. The isoelectric point (pI) refers to the pH at which a molecule has zero net charge in an electric field. The isoionic point, on the other hand, refers to the pH of a solution containing the molecule when it is in its purest form, without any added ions.
- Isoelectric Point (pI): pH at zero net charge in an electric field.
- Isoionic Point: pH of a pure solution of the molecule.
- Distinction: While similar, the isoionic point is a theoretical value, whereas the isoelectric point is experimentally measurable.
1.5. Importance of Isoelectric Point
The isoelectric point is a crucial parameter in biochemistry and molecular biology due to its impact on various properties of amino acids and proteins.
- Solubility: Proteins are least soluble at their pI because they tend to aggregate when there is no net charge to repel each other.
- Electrophoresis: Molecules at their pI do not migrate in an electric field, a property used in techniques like isoelectric focusing.
- Protein Purification: pI can be used to optimize conditions for protein purification techniques such as ion exchange chromatography.
- Biological Interactions: The charge state of a protein at a given pH affects its interactions with other molecules, including DNA, lipids, and other proteins.
2. Factors Influencing the Isoelectric Point
The isoelectric point is not a fixed value but is influenced by various factors, including the amino acid composition of the molecule, the presence of post-translational modifications, and environmental conditions such as temperature and ionic strength.
2.1. Amino Acid Composition
The primary determinant of a protein’s pI is its amino acid composition. Amino acids with ionizable side chains (e.g., aspartic acid, glutamic acid, lysine, arginine, histidine) contribute significantly to the overall charge of the protein and, consequently, its pI.
- Acidic Amino Acids (Asp, Glu): Lower the pI due to their negatively charged side chains at neutral pH.
- Basic Amino Acids (Lys, Arg, His): Raise the pI due to their positively charged side chains at neutral pH.
- Neutral Amino Acids: Have little impact on the pI.
2.2. Post-Translational Modifications
Post-translational modifications (PTMs) can alter the charge state of a protein and affect its pI. Common PTMs that influence pI include phosphorylation, glycosylation, and acetylation.
- Phosphorylation: Adds negatively charged phosphate groups, lowering the pI.
- Glycosylation: Adds carbohydrate moieties, which can increase or decrease the pI depending on the sugar composition.
- Acetylation: Neutralizes the positive charge of lysine residues, lowering the pI.
2.3. Environmental Conditions
Environmental conditions such as temperature, ionic strength, and the presence of specific ions can also influence the pI of a molecule.
- Temperature: Changes in temperature can alter the pKa values of ionizable groups, affecting the pI.
- Ionic Strength: High ionic strength can shield charges and affect the electrostatic interactions within the molecule, influencing the pI.
- Specific Ions: The presence of ions that bind to charged groups on the molecule can shift the pI.
2.4. Buffers and pH
The pH of the surrounding environment is the most direct influence on the charge state of a molecule. Buffers are often used to maintain a stable pH, and their choice can affect the apparent pI.
- Acidic Buffers (pH < pI): The molecule will be predominantly positively charged.
- Basic Buffers (pH > pI): The molecule will be predominantly negatively charged.
- Optimal Buffering: Choose a buffer with a pKa close to the desired pH to maintain stable conditions.
3. Calculating the Isoelectric Point
Calculating the isoelectric point involves determining the average of the relevant pKa values for the ionizable groups in the molecule. The method varies depending on whether the molecule has only amino and carboxyl groups or also contains ionizable side chains.
3.1. Amino Acids with Non-Ionizable Side Chains
For amino acids with non-ionizable side chains (e.g., glycine, alanine, valine), the pI is calculated by averaging the pKa values of the amino and carboxyl groups.
- Formula: pI = (pKa1 + pKa2) / 2
- Example: Glycine has pKa1 (carboxyl group) = 2.34 and pKa2 (amino group) = 9.60. Therefore, pI = (2.34 + 9.60) / 2 = 5.97.
3.2. Amino Acids with Acidic Side Chains
Amino acids with acidic side chains (e.g., aspartic acid, glutamic acid) have three pKa values: one for the carboxyl group, one for the amino group, and one for the side chain carboxyl group. To calculate the pI, average the two lowest pKa values.
- Formula: pI = (pKa1 + pKaR) / 2, where pKaR is the pKa of the acidic side chain.
- Example: Aspartic acid has pKa1 = 1.88 (α-carboxyl), pKa2 = 9.60 (amino group), and pKaR = 3.65 (side chain carboxyl). Therefore, pI = (1.88 + 3.65) / 2 = 2.77.
3.3. Amino Acids with Basic Side Chains
Amino acids with basic side chains (e.g., lysine, arginine, histidine) also have three pKa values: one for the carboxyl group, one for the amino group, and one for the side chain amino group. To calculate the pI, average the two highest pKa values.
- Formula: pI = (pKa2 + pKaR) / 2, where pKaR is the pKa of the basic side chain.
- Example: Lysine has pKa1 = 2.18 (carboxyl group), pKa2 = 8.95 (α-amino group), and pKaR = 10.53 (side chain amino). Therefore, pI = (8.95 + 10.53) / 2 = 9.74.
3.4. Proteins and Peptides
For proteins and peptides, the calculation of the pI is more complex because it involves considering the pKa values of all ionizable groups in the molecule. Several computational tools and databases can assist in predicting the pI of proteins based on their amino acid sequence.
- Approximation: Estimate pI by considering the relative abundance of acidic and basic amino acids.
- Computational Tools: Use software like ExPASy’s ProtParam tool to predict pI based on amino acid sequence.
- Experimental Methods: Determine pI experimentally using techniques like isoelectric focusing.
3.5. Titration Curves
Titration curves can be used to experimentally determine the pI of a molecule. By titrating an amino acid or protein with a strong acid or base, the buffering regions and equivalence points can be identified. The pI corresponds to the pH at the point where the molecule has no net charge.
- Method: Titrate the molecule with a strong acid or base.
- Analysis: Identify buffering regions and equivalence points.
- Determination: The pI is the pH at which the molecule has no net charge, usually the midpoint of the titration curve.
4. Experimental Determination of Isoelectric Point
The isoelectric point can be experimentally determined using various techniques, including electrophoresis and isoelectric focusing. These methods exploit the charge properties of molecules at different pH levels to separate and analyze them.
4.1. Electrophoresis
Electrophoresis is a technique used to separate molecules based on their size and charge by applying an electric field. At the pI, a molecule has no net charge and will not migrate in the electric field.
- Principle: Separate molecules based on size and charge.
- Procedure: Apply an electric field to a sample at different pH levels.
- Determination: The pI is the pH at which the molecule does not migrate.
4.2. Isoelectric Focusing (IEF)
Isoelectric focusing is a high-resolution electrophoretic technique that separates proteins based on their pI. A pH gradient is established in a gel, and proteins migrate until they reach the pH corresponding to their pI, where they stop migrating and become focused into a narrow band.
- Principle: Separate proteins based on their pI in a pH gradient.
- Procedure: Establish a pH gradient in a gel and apply an electric field.
- Determination: Proteins migrate to the pH corresponding to their pI and form sharp bands.
4.3. Capillary Isoelectric Focusing (cIEF)
Capillary isoelectric focusing is an automated version of IEF that is performed in a capillary tube. This technique offers high resolution, sensitivity, and reproducibility, making it suitable for analyzing complex protein mixtures.
- Principle: Automated version of IEF performed in a capillary tube.
- Advantages: High resolution, sensitivity, and reproducibility.
- Applications: Analyzing complex protein mixtures and determining protein purity.
4.4. Two-Dimensional Gel Electrophoresis (2D-PAGE)
Two-dimensional gel electrophoresis combines IEF and SDS-PAGE to separate proteins based on their pI and molecular weight. Proteins are first separated by IEF in one dimension and then by SDS-PAGE in the second dimension, providing high-resolution separation of complex protein mixtures.
- Principle: Combine IEF and SDS-PAGE to separate proteins based on pI and molecular weight.
- Procedure: Separate proteins by IEF in the first dimension and by SDS-PAGE in the second dimension.
- Applications: Proteomics research, analyzing complex protein mixtures, and identifying protein isoforms.
4.5. Factors Affecting Experimental pI Measurement
Several factors can affect the accuracy of experimental pI measurements, including buffer composition, temperature, and the presence of contaminants. It is important to carefully control these variables to obtain reliable results.
- Buffer Composition: The choice of buffer can affect the pH gradient and the migration of proteins.
- Temperature: Temperature can influence the pKa values of ionizable groups and the mobility of proteins.
- Contaminants: The presence of salts, detergents, or other contaminants can interfere with the separation and focusing of proteins.
5. Applications of Isoelectric Point
The isoelectric point has numerous applications in biochemistry, biotechnology, and medicine. Understanding and manipulating the pI of molecules is crucial in various techniques, including protein purification, drug delivery, and diagnostics.
5.1. Protein Purification
The pI is a key parameter in protein purification techniques, such as ion exchange chromatography and isoelectric precipitation. By adjusting the pH of the solution, proteins can be selectively bound to or eluted from chromatographic resins.
- Ion Exchange Chromatography: Proteins are separated based on their charge properties.
- Isoelectric Precipitation: Proteins are precipitated at their pI due to reduced solubility.
- Affinity Chromatography: Proteins are purified based on specific binding interactions.
5.2. Drug Delivery
The pI of drug-carrying nanoparticles can be tuned to optimize their delivery to specific tissues or cells. Positively charged nanoparticles (high pI) tend to interact more strongly with negatively charged cell membranes, facilitating cellular uptake.
- Targeted Delivery: Nanoparticles are designed to target specific cells or tissues.
- Controlled Release: Drugs are released at a controlled rate to maintain therapeutic levels.
- Improved Bioavailability: Drugs are formulated to enhance their absorption and distribution.
5.3. Diagnostics
Isoelectric focusing and 2D-PAGE are used in diagnostics to identify protein biomarkers for various diseases. Changes in the pI or abundance of specific proteins can indicate the presence or progression of a disease.
- Biomarker Discovery: Identify proteins that are indicative of a disease state.
- Disease Monitoring: Track changes in protein levels to monitor disease progression.
- Personalized Medicine: Tailor treatment strategies based on individual protein profiles.
5.4. Food Science
In food science, the pI is important for understanding the behavior of proteins in food products. It affects the texture, stability, and solubility of proteins, influencing the quality and shelf life of foods.
- Texture Modification: Adjusting the pH to alter the texture of food products.
- Protein Solubility: Optimizing conditions for protein extraction and processing.
- Emulsion Stability: Enhancing the stability of emulsions by controlling protein charge.
5.5. Environmental Science
The pI is used in environmental science to study the behavior of pollutants and microorganisms in soil and water. Understanding the charge properties of these substances helps in developing effective remediation strategies.
- Pollutant Removal: Designing materials to selectively bind and remove pollutants.
- Microbial Activity: Studying the interactions between microorganisms and their environment.
- Water Treatment: Optimizing processes for water purification and disinfection.
6. Isoelectric Point and pH: A Detailed Comparison
Understanding the relationship between the isoelectric point and pH is crucial for predicting the behavior of molecules in solution. The pH of the environment determines the charge state of a molecule relative to its pI.
6.1. pH Below the pI
When the pH of the solution is below the pI, the molecule will be protonated and carry a net positive charge. This is because the concentration of protons (H+) is high, favoring the protonation of ionizable groups.
- Charge State: Net positive charge.
- Protonation: Ionizable groups are protonated.
- Behavior: Molecules tend to migrate towards the cathode in electrophoresis.
6.2. pH Above the pI
When the pH of the solution is above the pI, the molecule will be deprotonated and carry a net negative charge. This is because the concentration of hydroxide ions (OH-) is high, favoring the deprotonation of ionizable groups.
- Charge State: Net negative charge.
- Deprotonation: Ionizable groups are deprotonated.
- Behavior: Molecules tend to migrate towards the anode in electrophoresis.
6.3. pH at the pI
At the isoelectric point (pH = pI), the molecule carries no net charge. The positive and negative charges are balanced, resulting in minimal migration in an electric field and often reduced solubility.
- Charge State: Net zero charge.
- Equilibrium: Positive and negative charges are balanced.
- Behavior: Minimal migration in an electric field and reduced solubility.
6.4. Buffering Capacity
Molecules exhibit buffering capacity around their pKa values, which are the pH values at which an ionizable group is half-protonated and half-deprotonated. The buffering capacity is highest when the pH is close to the pKa, allowing the molecule to resist changes in pH upon the addition of acid or base.
- Definition: The ability to resist changes in pH.
- Mechanism: Occurs when the concentration of an acid and its conjugate base are equal.
- Importance: Maintains stable pH conditions in biological systems.
6.5. Physiological Relevance
The pI and pH are critical in physiological systems, affecting protein-protein interactions, enzyme activity, and cellular processes. Maintaining proper pH levels is essential for the proper functioning of biological systems.
- Enzyme Activity: Enzymes have optimal activity at specific pH levels.
- Protein Interactions: Protein-protein interactions are influenced by charge complementarity.
- Cellular Processes: pH affects membrane transport, signal transduction, and other cellular functions.
7. Advanced Topics in Isoelectric Point
Delving deeper into the concept of the isoelectric point reveals more complex aspects, including the effects of protein folding, the use of computational tools for pI prediction, and the role of pI in protein engineering.
7.1. Effects of Protein Folding
The three-dimensional structure of a protein can influence its pI by affecting the accessibility and pKa values of ionizable groups. Buried residues may have different pKa values compared to surface residues due to changes in their microenvironment.
- Conformation: The three-dimensional structure of the protein.
- Microenvironment: The local chemical environment surrounding ionizable groups.
- Accessibility: The extent to which ionizable groups are exposed to the solvent.
7.2. Computational Tools for pI Prediction
Several computational tools are available for predicting the pI of proteins based on their amino acid sequence. These tools use algorithms that consider the pKa values of all ionizable groups and can provide accurate pI predictions for a wide range of proteins.
- ExPASy ProtParam: A tool for calculating various protein parameters, including pI.
- অন্যান্য tools: Several other tools are available for pI prediction, each with its own strengths and limitations.
- Accuracy: The accuracy of pI predictions depends on the quality of the input sequence and the algorithms used.
7.3. pI in Protein Engineering
Protein engineering involves modifying the amino acid sequence of a protein to alter its properties. The pI can be a target for protein engineering to improve protein solubility, stability, or binding affinity.
- Mutagenesis: Introducing mutations to alter the amino acid sequence.
- Rational Design: Designing mutations based on structural and functional considerations.
- Directed Evolution: Selecting for mutations that improve the desired properties.
7.4. Isoelectric Point and Colloid Stability
The isoelectric point is important in colloid chemistry for understanding the stability of colloidal dispersions. Colloids are most stable when their particles have a high surface charge, which prevents aggregation. At the pI, the surface charge is minimal, leading to particle aggregation and instability.
- Colloidal Dispersions: Mixtures of particles dispersed in a continuous medium.
- Surface Charge: The electrical charge on the surface of particles.
- Stability: The ability of a colloidal dispersion to resist aggregation.
7.5. Future Directions in pI Research
Future research on the isoelectric point will likely focus on developing more accurate pI prediction methods, exploring the role of pI in complex biological systems, and using pI as a tool for designing novel biomaterials and therapeutics.
- Improved Prediction Methods: Developing more accurate algorithms for pI prediction.
- Biological Systems: Studying the role of pI in complex cellular processes.
- Biomaterials: Designing novel materials with tailored pI properties.
8. Practical Examples and Case Studies
To illustrate the practical significance of the isoelectric point, let’s examine a few case studies in different fields.
8.1. Case Study: Protein Purification of Insulin
Insulin is a hormone that regulates blood glucose levels. Its purification from pancreatic extracts involves multiple steps, including isoelectric precipitation. Insulin has a pI of 5.4, so adjusting the pH to this value causes insulin to precipitate out of the solution, allowing it to be separated from other proteins.
- Objective: Purify insulin from pancreatic extracts.
- Method: Adjust the pH to insulin’s pI (5.4) to induce precipitation.
- Outcome: Selective precipitation of insulin, facilitating its purification.
8.2. Case Study: Drug Delivery of Nanoparticles
Researchers have developed nanoparticles with a pI of 6.5 for targeted drug delivery to cancer cells. At the slightly acidic pH of the tumor microenvironment, these nanoparticles become positively charged, enhancing their interaction with the negatively charged cell membranes of cancer cells, leading to increased drug uptake.
- Objective: Deliver drugs specifically to cancer cells.
- Method: Design nanoparticles with a pI that becomes positively charged in the tumor microenvironment.
- Outcome: Enhanced drug uptake by cancer cells due to increased electrostatic interactions.
8.3. Case Study: Diagnostic Assay for Alzheimer’s Disease
A diagnostic assay for Alzheimer’s disease involves measuring the pI of amyloid-beta peptides in cerebrospinal fluid (CSF). Changes in the pI of these peptides can indicate the presence of post-translational modifications associated with the disease, allowing for early detection.
- Objective: Develop a diagnostic assay for Alzheimer’s disease.
- Method: Measure the pI of amyloid-beta peptides in CSF.
- Outcome: Early detection of Alzheimer’s disease based on changes in peptide pI.
8.4. Case Study: Food Processing of Soy Protein
Soy protein is widely used in food products as a plant-based protein source. Understanding its pI (around 4.5) is crucial for optimizing its extraction and processing. Adjusting the pH to the pI can cause soy protein to precipitate, facilitating its separation from other components and improving its texture and functionality in food applications.
- Objective: Optimize the extraction and processing of soy protein.
- Method: Adjust the pH to soy protein’s pI (4.5) to induce precipitation.
- Outcome: Improved separation and functionality of soy protein in food products.
8.5. Case Study: Environmental Remediation of Heavy Metals
Researchers are using modified microorganisms with a high pI to remove heavy metals from contaminated water. The positively charged cell surfaces of these microorganisms attract negatively charged heavy metal ions, facilitating their removal from the water.
- Objective: Remove heavy metals from contaminated water.
- Method: Use microorganisms with a high pI to attract heavy metal ions.
- Outcome: Effective removal of heavy metals from contaminated water.
9. Common Misconceptions About Isoelectric Point
Several misconceptions exist regarding the isoelectric point. Addressing these misunderstandings is crucial for a comprehensive understanding of the concept.
9.1. Misconception: pI is a Fixed Value
The pI is often mistakenly considered a fixed value for a given molecule. In reality, the pI can be influenced by environmental conditions, post-translational modifications, and interactions with other molecules.
- Reality: The pI can vary depending on environmental conditions and modifications.
- Factors: Temperature, ionic strength, post-translational modifications, and interactions with other molecules.
9.2. Misconception: Molecules are Completely Uncharged at the pI
It is a common misconception that molecules are completely uncharged at their pI. In fact, molecules at their pI have an equal balance of positive and negative charges, resulting in a net charge of zero, but they still possess individual charges.
- Reality: Molecules have an equal balance of positive and negative charges, resulting in a net charge of zero.
- Individual Charges: Molecules still possess individual charges that balance each other out.
9.3. Misconception: pI is Only Relevant for Amino Acids and Proteins
While the pI is commonly discussed in the context of amino acids and proteins, it is relevant for any molecule with ionizable groups, including nucleic acids, lipids, and synthetic polymers.
- Reality: The pI is relevant for any molecule with ionizable groups.
- Examples: Nucleic acids, lipids, synthetic polymers, and other amphoteric molecules.
9.4. Misconception: pI is the Same as Neutral pH
The pI is often confused with neutral pH (7.0). However, the pI is a specific property of a molecule and depends on its chemical structure, while neutral pH is a property of the solution.
- Reality: The pI depends on the chemical structure of the molecule and may not be 7.0.
- Neutral pH: Neutral pH is a property of the solution, where the concentration of H+ and OH- ions is equal.
9.5. Misconception: pI Always Correlates with Solubility
While molecules are generally least soluble at their pI, this is not always the case. Other factors, such as salt concentration, temperature, and the presence of specific ions, can also affect solubility.
- Reality: Other factors can influence solubility.
- Additional Factors: Salt concentration, temperature, and the presence of specific ions.
10. Frequently Asked Questions (FAQ) About Isoelectric Point
Here are some frequently asked questions about the isoelectric point, along with detailed answers.
- What is the significance of knowing the pI of a protein?
Understanding the pI of a protein is critical for various applications, including protein purification, electrophoresis, and drug delivery. It helps predict the protein’s behavior under different pH conditions and optimize experimental procedures. - How does the pI affect protein solubility?
Proteins are generally least soluble at their pI because they tend to aggregate when there is no net charge to repel each other. At pH values away from the pI, the net charge increases, enhancing solubility. - Can the pI of a protein be altered?
Yes, the pI of a protein can be altered through post-translational modifications, such as phosphorylation, glycosylation, or by introducing mutations to change its amino acid composition. - What is the difference between pKa and pI?
pKa is the pH at which half of the molecules of a specific ionizable group are protonated, while pI is the pH at which the entire molecule has a net charge of zero. - How is isoelectric focusing used in protein analysis?
Isoelectric focusing separates proteins based on their pI by applying an electric field to a pH gradient. Proteins migrate until they reach the pH corresponding to their pI, where they stop migrating, allowing for high-resolution separation. - What are some real-world applications of the pI concept?
Real-world applications include protein purification, drug delivery, diagnostics, food science, and environmental science. The pI is used to optimize conditions for these processes and improve their effectiveness. - How do acidic and basic amino acids affect the pI of a protein?
Acidic amino acids lower the pI of a protein, while basic amino acids raise the pI. The overall pI depends on the relative abundance and distribution of these amino acids. - What is the role of buffers in determining the pI?
Buffers are used to maintain a stable pH during experiments. The choice of buffer can affect the apparent pI, so it is important to select a buffer with a pKa close to the desired pH. - How can I predict the pI of a protein using computational tools?
Several computational tools, such as ExPASy’s ProtParam tool, can predict the pI of a protein based on its amino acid sequence. These tools use algorithms that consider the pKa values of all ionizable groups. - Why is it important to control the pH in protein experiments?
Controlling the pH is critical in protein experiments because it affects the charge state of the protein, influencing its solubility, stability, and interactions with other molecules. Maintaining proper pH levels is essential for obtaining reliable results.
Understanding the isoelectric point and its relationship to pH is essential for anyone working in biochemistry, molecular biology, or related fields. By grasping the concepts discussed in this article, you can better predict and manipulate the behavior of molecules in various applications.
Are you looking for more in-depth comparisons and detailed analyses to aid your decision-making? Visit COMPARE.EDU.VN today and explore a wide range of topics to help you make informed choices. Our comprehensive comparisons are designed to provide you with the clarity and confidence you need.
Ready to make smarter choices? Head over to COMPARE.EDU.VN now Address: 333 Comparison Plaza, Choice City, CA 90210, United States. Whatsapp: +1 (626) 555-9090. Trang web: compare.edu.vn