Comparing ionic radii is essential for understanding the chemical properties and behavior of elements. Ionic radius, the radius of an ion in an ionic crystal, influences various aspects of chemistry, from lattice energy to the arrangement of ions in crystal structures. At COMPARE.EDU.VN, we provide comprehensive comparisons to help you make informed decisions. This article explores the trends and factors affecting ionic radii, offering a detailed guide for students, consumers, and experts alike. Discover the ease and accuracy of comparing ionic radii with COMPARE.EDU.VN and delve into effective nuclear charge, isoelectronic series, and periodic trends to gain a thorough understanding.
1. Understanding Atomic and Ionic Radii
1.1 Defining Atomic Radii
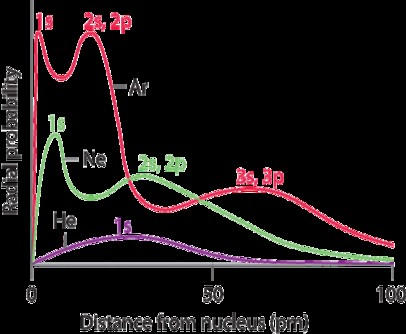
Atomic radius is the distance from the nucleus to the outermost electron shell of an atom. It’s a fundamental property that affects how atoms interact and form chemical bonds. However, defining a precise atomic radius is challenging due to the probabilistic nature of electron locations.
1.2 Methods for Measuring Atomic Radii
Chemists use several methods to determine atomic radii:
- Covalent Atomic Radius (r_cov): Half the distance between the nuclei of two identical atoms joined by a covalent bond in the same molecule.
- Metallic Atomic Radius (r_met): Half the distance between the nuclei of two adjacent atoms in a pure solid metal.
- Van der Waals Atomic Radius (r_vdW): Half the internuclear distance between two non-bonded atoms in a solid.
1.3 Defining Ionic Radii
Ionic radius refers to the radius of an ion in an ionic crystal. Ions are formed when atoms gain or lose electrons, resulting in charged particles. Cations are positively charged ions formed by losing electrons, while anions are negatively charged ions formed by gaining electrons.
2. Factors Affecting Ionic Radius
2.1 Nuclear Charge
The nuclear charge, or the number of protons in the nucleus, significantly impacts ionic radius. A higher nuclear charge attracts electrons more strongly, pulling them closer to the nucleus and reducing the ion’s size.
2.2 Number of Electrons
The number of electrons also affects ionic radius. More electrons increase electron-electron repulsion, causing the electron cloud to expand and the ionic radius to increase.
2.3 Electron Shielding
Electron shielding refers to the ability of inner electrons to reduce the effective nuclear charge experienced by outer electrons. Increased shielding decreases the attraction between the nucleus and outer electrons, leading to larger ionic radii.
2.4 Effective Nuclear Charge (Zeff)
The effective nuclear charge is the net positive charge experienced by an electron in a multi-electron atom. It is the actual nuclear charge minus the shielding effect of inner electrons. A higher effective nuclear charge results in a smaller ionic radius.
3. Periodic Trends in Ionic Radii
3.1 Trends Across a Period
Across a period (from left to right) in the periodic table, ionic radius generally decreases for isoelectronic species (ions with the same number of electrons). This is because the nuclear charge increases, leading to a stronger attraction and a smaller size.
3.2 Trends Down a Group
Down a group (from top to bottom) in the periodic table, ionic radius increases. This is primarily due to the addition of electron shells, which increases the distance between the nucleus and the outermost electrons.
4. Comparing Cations and Anions
4.1 Cations vs. Neutral Atoms
Cations are always smaller than their parent neutral atoms. When an atom loses electrons to form a cation, electron-electron repulsion decreases, and the effective nuclear charge increases, causing the ion to shrink.
4.2 Anions vs. Neutral Atoms
Anions are always larger than their parent neutral atoms. When an atom gains electrons to form an anion, electron-electron repulsion increases, and the effective nuclear charge decreases, leading to an expansion of the electron cloud.
5. Isoelectronic Series
5.1 Definition of Isoelectronic Series
An isoelectronic series is a group of ions or atoms that have the same number of electrons but different nuclear charges. Comparing the ionic radii within an isoelectronic series provides valuable insights into the effect of nuclear charge on ionic size.
5.2 Trends in Isoelectronic Series
In an isoelectronic series, ionic radius decreases as the nuclear charge increases. For example, consider the isoelectronic series with the neon closed-shell configuration (1s22s22p6): N3-, O2-, F-, Na+, Mg2+, and Al3+. As the nuclear charge increases from +7 (N) to +13 (Al), the ionic radius decreases.
6. Examples of Ionic Radius Comparison
6.1 Comparing Na+ and Cl- in NaCl
In sodium chloride (NaCl), the internuclear distance between Na+ and Cl- can be measured to determine their ionic radii. The measured distance is the sum of the radii of the cation and anion.
6.2 Comparing Different Oxidation States of the Same Element
Elements with multiple oxidation states exhibit different ionic radii. For example, iron can form Fe2+ and Fe3+ ions. Fe3+ has a smaller ionic radius than Fe2+ because it has a higher positive charge and fewer electrons.
7. Practical Applications of Understanding Ionic Radius
7.1 Predicting Lattice Energy
Ionic radius is crucial in predicting the lattice energy of ionic compounds. Lattice energy is the energy required to separate one mole of a solid ionic compound into gaseous ions. Smaller ions with higher charges result in higher lattice energies due to stronger electrostatic attractions.
7.2 Understanding Crystal Structures
Ionic radii influence the arrangement of ions in crystal structures. The relative sizes of cations and anions determine the coordination number and the overall structure of the crystal lattice.
7.3 Designing New Materials
Knowledge of ionic radii is essential in designing new materials with specific properties. By selecting ions with appropriate sizes and charges, materials scientists can tailor the physical and chemical characteristics of materials for various applications.
8. Tools and Resources for Comparing Ionic Radii
8.1 Online Databases
Several online databases provide comprehensive information on ionic radii, including values for various elements and oxidation states. These databases are valuable resources for researchers, students, and anyone interested in comparing ionic sizes. Web Elements [www.webelements.com]. Web Elements is an excellent online source for looking up atomic properties.
8.2 Periodic Table Trends
Using the periodic table as a reference can help predict relative ionic sizes. Understanding the trends across periods and down groups allows for quick comparisons without needing exact values.
8.3 COMPARE.EDU.VN: Your Comparison Resource
At COMPARE.EDU.VN, we offer a platform for comparing ionic radii and other chemical properties. Our tools provide detailed comparisons, helping users make informed decisions based on reliable data. Visit our website to explore and compare a wide range of chemical properties.
9. Case Studies
9.1 Comparing Halide Ions
The halide ions (F-, Cl-, Br-, I-) provide a clear example of the trend in ionic radii down a group. As the number of electron shells increases from fluorine to iodine, the ionic radius increases accordingly.
9.2 Comparing Alkali Metal Ions
The alkali metal ions (Li+, Na+, K+, Rb+, Cs+) also illustrate the trend of increasing ionic radius down a group. Cesium ion (Cs+) is significantly larger than lithium ion (Li+) due to the additional electron shells.
10. Common Mistakes to Avoid When Comparing Ionic Radii
10.1 Neglecting the Charge of the Ion
Failing to consider the charge of the ion is a common mistake. Ions with higher positive charges are smaller, while ions with higher negative charges are larger. Always account for the charge when comparing ionic radii.
10.2 Ignoring Isoelectronic Series
When comparing ions with the same number of electrons, remember to consider the nuclear charge. Ignoring isoelectronic series can lead to incorrect conclusions about relative sizes.
10.3 Overlooking Shielding Effects
Electron shielding significantly impacts the effective nuclear charge. Overlooking these effects can result in inaccurate predictions about ionic radii.
11. Advanced Concepts in Ionic Radius
11.1 Polarization Effects
Polarization refers to the distortion of the electron cloud of an ion by the presence of a nearby ion with a high charge density. Polarization effects can influence ionic radii and the properties of ionic compounds.
11.2 Covalency Effects
In some ionic compounds, there is a degree of covalency in the bonding. Covalency effects can alter the electron distribution and affect the apparent ionic radii.
12. Latest Research and Developments
12.1 New Methods for Measuring Ionic Radii
Researchers are continuously developing new and more accurate methods for measuring ionic radii. These methods provide more precise data for understanding the properties of materials.
12.2 Applications in Nanotechnology
Ionic radii play a crucial role in nanotechnology, where the properties of materials are highly dependent on the size and charge of ions. Recent advances in nanotechnology are leveraging ionic radii to create new devices and materials with unique characteristics.
13. Expert Opinions on Ionic Radius
13.1 Insights from Leading Chemists
Leading chemists emphasize the importance of understanding ionic radii for predicting the behavior of chemical compounds. They highlight the need for accurate data and a thorough understanding of the factors affecting ionic size.
13.2 Future Directions in Research
Experts suggest that future research should focus on developing more sophisticated models for predicting ionic radii and exploring the applications of ionic size in designing new materials.
14. Conclusion
Understanding how to compare ionic radii is essential for anyone studying chemistry, materials science, or related fields. By considering the factors affecting ionic size and following periodic trends, you can make accurate predictions and gain valuable insights into the properties of chemical compounds. For reliable comparisons and detailed information, visit COMPARE.EDU.VN.
15. Call to Action
Ready to dive deeper into comparing ionic radii? Visit COMPARE.EDU.VN today to explore our comprehensive comparison tools and make informed decisions. Whether you’re a student, a consumer, or an expert, COMPARE.EDU.VN provides the resources you need. Contact us at 333 Comparison Plaza, Choice City, CA 90210, United States, or via Whatsapp at +1 (626) 555-9090.
16. Frequently Asked Questions (FAQ)
16.1 What is ionic radius?
Ionic radius is the radius of an ion in an ionic crystal, which affects the arrangement and properties of the crystal structure.
16.2 How is ionic radius measured?
Ionic radius is typically determined by measuring the distance between the nuclei of ions in an ionic compound and then apportioning this distance based on theoretical calculations or experimental data.
16.3 Why are cations smaller than their parent atoms?
Cations are smaller because they lose electrons, reducing electron-electron repulsion and increasing the effective nuclear charge, which pulls the remaining electrons closer to the nucleus.
16.4 Why are anions larger than their parent atoms?
Anions are larger because they gain electrons, increasing electron-electron repulsion and decreasing the effective nuclear charge, which allows the electron cloud to expand.
16.5 What is an isoelectronic series?
An isoelectronic series is a group of ions or atoms that have the same number of electrons but different nuclear charges.
16.6 How does nuclear charge affect ionic radius in an isoelectronic series?
In an isoelectronic series, as the nuclear charge increases, the ionic radius decreases because the greater positive charge pulls the electrons closer to the nucleus.
16.7 What are the periodic trends in ionic radii?
Ionic radius generally decreases across a period (for isoelectronic species) and increases down a group in the periodic table.
16.8 How does electron shielding affect ionic radius?
Electron shielding reduces the effective nuclear charge experienced by outer electrons, leading to larger ionic radii.
16.9 What is the effective nuclear charge (Zeff)?
The effective nuclear charge is the net positive charge experienced by an electron in a multi-electron atom, accounting for the shielding effect of inner electrons.
16.10 Where can I find reliable data for comparing ionic radii?
You can find reliable data and comparison tools at compare.edu.vn, as well as in online databases and chemistry textbooks.