Comparing atomic radius can be complex, but COMPARE.EDU.VN simplifies the process. Understanding how atomic radius changes across the periodic table and the factors influencing these trends is crucial in chemistry. This guide explores the definition of atomic radius, methods for its determination, periodic trends, and comparison of ionic radii, offering a comprehensive understanding of this essential concept.
1. Defining Atomic Radius
Atoms and ions are not hard spheres with definite boundaries. The quantum mechanical model describes electron density diminishing gradually with distance from the nucleus. This makes it challenging to define an exact size. However, relative atomic sizes are crucial in chemistry.
1.1. The Challenge of Measurement
Electron density diminishes with increasing distance from the nucleus, making it difficult to define a clear boundary.
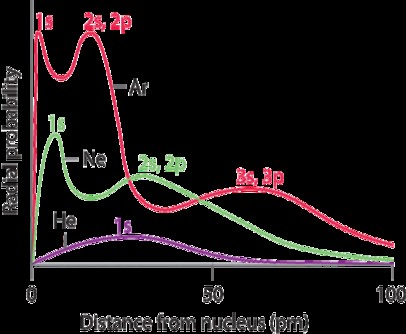
Alt Text: Radial probability plots for Helium, Neon, and Argon, showing electron density peaks.
This figure illustrates how electron density peaks at different distances for each element, corresponding to electron density in principal shells.
1.2. Covalent Atomic Radius
Chemists measure distances between nuclei in covalently bonded atoms to estimate atomic sizes. The covalent atomic radius is half the distance between the nuclei of two identical atoms joined by a covalent bond in the same molecule.
1.3. Metallic Atomic Radius
For metals, the metallic atomic radius is defined as half the distance between the nuclei of two adjacent metal atoms in the solid.
1.4. Van der Waals Atomic Radius
The van der Waals atomic radius is half the internuclear distance between two nonbonded atoms in the solid, useful for elements like noble gases that don’t form stable compounds.
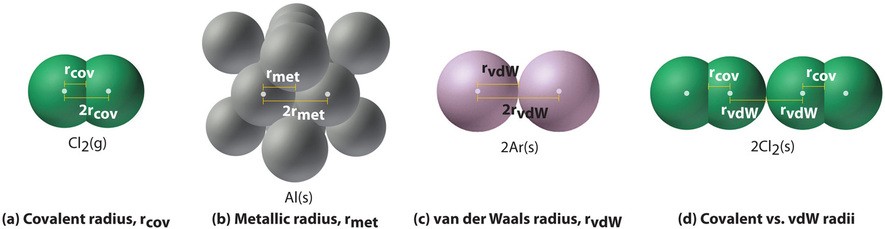
Alt Text: Illustration of covalent, metallic, and van der Waals radii.
This image contrasts covalent versus van der Waals radii of chlorine.
2. Methods for Determining Atomic Radius
Since direct measurement is impossible, chemists use various methods to calculate atomic radii.
2.1. Experimental Methods
Experimental techniques include X-ray diffraction for crystalline solids, which measures distances between atomic nuclei.
2.2. Quantum Mechanical Calculations
Quantum mechanical functions provide a way to calculate atomic radii, comparing intrinsic sizes across elements and revealing periodic variations.
2.3. Self-Consistent Calculations
Chemists have developed self-consistent methods using quantum mechanical functions to calculate atomic radii. These calculations provide a means to compare the intrinsic sizes of all elements and highlight periodic trends.
3. Periodic Trends in Atomic Radii
Atomic size varies periodically in the periodic table.
3.1. Trends Across a Period
Atomic radii decrease from left to right across a row. This is due to increasing effective nuclear charge ((Z{eff})), the net positive charge experienced by valence electrons. Greater (Z{eff}) attracts outermost electrons more strongly, reducing atomic radius.
3.2. Trends Down a Group
Atomic radii increase from top to bottom down a column. As the principal quantum number (n) increases, orbitals become larger, even though the nuclear charge increases.
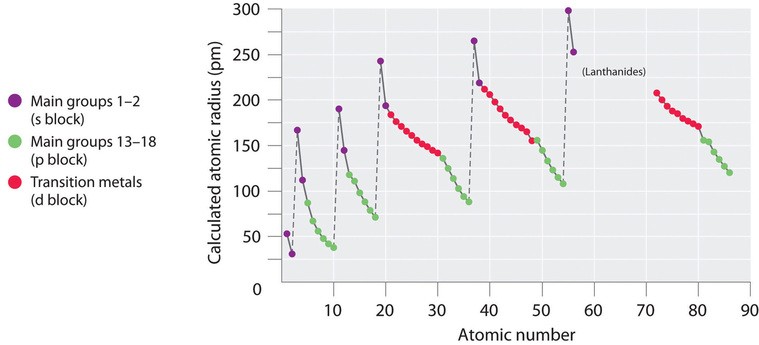
Alt Text: Periodic variation of atomic radius with atomic number.
This plot highlights the decrease in atomic radii across a period and the increase down a group.
3.3. Effective Nuclear Charge
Differences in effective nuclear charges ((Z_{eff})) experienced by electrons in outermost orbitals drive atomic size trends. Shielding effects, where inner electrons reduce the nuclear charge experienced by outer electrons, are crucial.
4. Understanding Electron Shielding
Electron shielding significantly affects atomic radii trends.
4.1. Shielding Effects
Inner electrons shield outer electrons from the full nuclear charge, reducing the effective nuclear charge experienced by valence electrons.
4.2. Ineffective Shielding
Electrons in the same principal shell are less effective at shielding each other from nuclear charge compared to inner shell electrons.
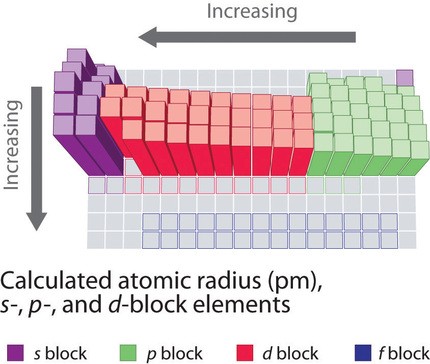
Alt Text: Atomic radii of s-, p-, and d-block elements.
This image illustrates relative atomic sizes based on quantum mechanical wave functions.
4.3. Impact on Atomic Size
Effective shielding causes atoms to be larger than expected, especially down a group where the principal quantum number increases.
5. Comparing Atomic Radii of Specific Elements
Let’s compare atomic radii of specific elements to understand the trends better.
5.1. Comparing Across the Second Row
Elements in the second row (Li to Ne) have a filled (1s^2) inner shell. As nuclear charge increases from +3 to +10, electrons are added to 2s and 2p orbitals. These electrons do not effectively shield each other, leading to increased effective nuclear charge and decreased atomic size.
5.2. Comparing Down Group 1
In Group 1, atomic size increases significantly down the column. While the principal quantum number n increases from 2 to 6, the nuclear charge increases from +3 to +55.
5.3. Cesium vs. Lithium
Cesium ([Xe](6s^1)) is much larger than lithium ((2s^1)) because the effective nuclear charge experienced by cesium’s outermost electrons is much less than expected due to shielding by filled inner shells.
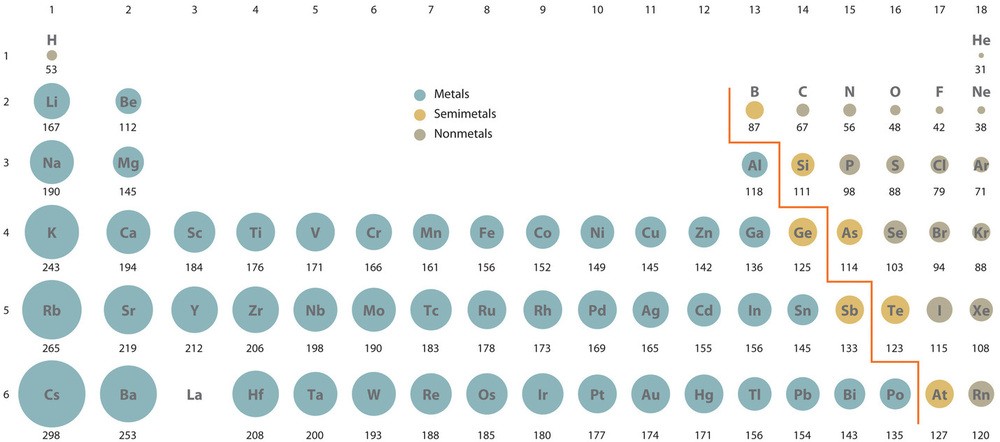
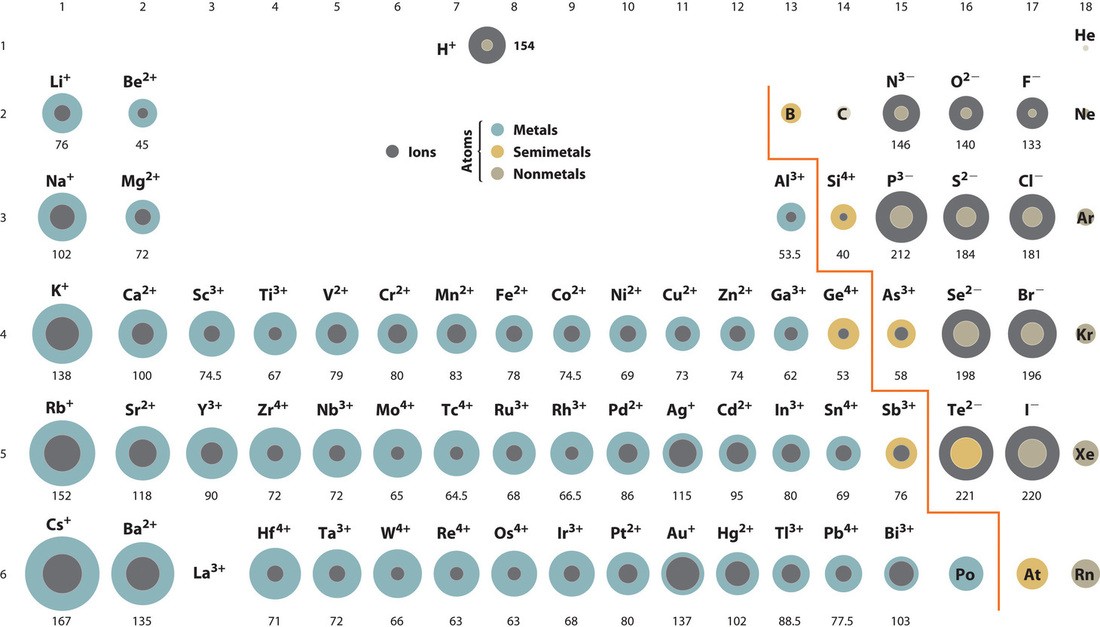
Alt Text: Atomic radius of elements across the periodic table.
The image shows that atomic radius increases from right to left across a period and down a group.
6. Ionic Radii and Isoelectronic Series
Ions are formed by either adding or removing electrons from neutral atoms. Understanding ionic radii helps to compare sizes of ions and atoms.
6.1. Formation of Ions
Cations (positive ions) form when electrons are removed, while anions (negative ions) form when electrons are added.
6.2. Defining Ionic Radius
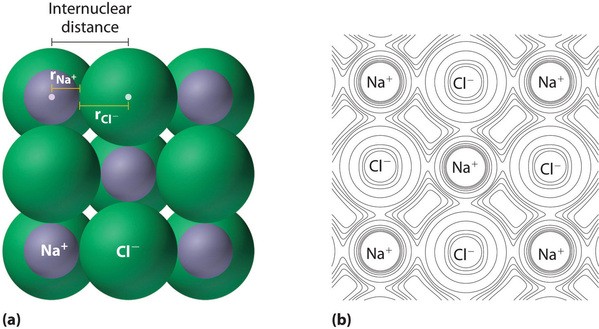
Ionic radius is the radius of a cation or anion. It is determined by measuring the distance between the nuclei of a cation and an adjacent anion in an ionic compound.
Alt Text: Ionic radius defined in an ionic compound.
This figure illustrates how internuclear distance corresponds to the sum of radii of cation and anion.
6.3. Comparing Ionic and Atomic Radii
Cations are always smaller than their parent neutral atoms because electron repulsion decreases, and effective nuclear charge increases. Anions are always larger than their parent neutral atoms due to increased electron-electron repulsion and decreased effective nuclear charge.
6.4. Trends in Ionic Radii
Ionic radii follow the same vertical trend as atomic radii. For ions with the same charge, ionic radius increases down a column.
7. Isoelectronic Series
An isoelectronic series consists of species with the same number of electrons but different nuclear charges.
7.1. Definition and Examples
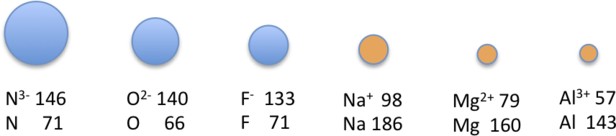
For example, (N^{3-}), (O^{2-}), (F^{-}), (Na^{+}), (Mg^{2+}), and (Al^{3+}) all have the neon closed-shell configuration ((1s^22s^22p^6)).
7.2. Trends in Isoelectronic Series
In an isoelectronic series, the size of ions decreases as nuclear charge increases. Greater positive charge attracts electrons more strongly, reducing the radius.
Alt Text: Illustration of an Isoelectronic series.
This image shows how ionic radius decreases with increasing atomic number in the isoelectronic series.
7.3. Impact of Nuclear Charge
The ion with the greatest nuclear charge is the smallest, while the ion with the smallest nuclear charge is the largest in an isoelectronic series.
8. Factors Influencing Atomic Radius
Several factors influence atomic radius, making the topic complex.
8.1. Nuclear Charge
Greater nuclear charge pulls electrons closer to the nucleus, reducing atomic radius.
8.2. Shielding Effect
Increased shielding reduces the effective nuclear charge, leading to larger atomic radius.
8.3. Principal Quantum Number
Higher principal quantum numbers mean electrons occupy larger orbitals, increasing atomic radius.
9. Examples of Comparing Atomic Radii
Comparing specific elements and ions can clarify the principles.
9.1. Example 1: Aluminum, Carbon, and Silicon
Arrange aluminum, carbon, and silicon in order of increasing atomic radius. Carbon and silicon are in Group 14, with carbon above silicon, so C < Si. Aluminum and silicon are in the third row, with aluminum to the left, so Si < Al. Combining gives C < Si < Al.
9.2. Example 2: Oxygen, Phosphorus, Potassium, and Sulfur
Arrange oxygen, phosphorus, potassium, and sulfur in order of increasing size. The answer is O < S < P < K.
9.3. Sodium Ion Radii
The radius of sodium varies in its different oxidation states:
| Na+ | Na0 | Na- | |
|---|---|---|---|
| Configuration | (1s^22s^22p^6) | (1s^22s^22p^63s^1) | (1s^22s^22p^63s^2) |
| Radius (pm) | 102 | 154 | 202 |
Alt Text: Ionic radii of common ionic states of elements.
This figure shows the relative sizes of neutral atoms and their ions.
10. Advanced Concepts in Atomic Radius
Exploring advanced concepts provides a deeper understanding of atomic radii.
10.1. Relativistic Effects
For very heavy elements, relativistic effects can influence orbital energies and sizes, leading to unexpected trends in atomic radii.
10.2. Lanthanide Contraction
The lanthanide contraction refers to the decrease in atomic and ionic radii of the lanthanide elements with increasing atomic number.
10.3. Applications of Atomic Radius
Understanding atomic radii is crucial in various fields, including materials science, drug design, and catalysis.
11. Practical Applications of Atomic Radius Knowledge
The understanding of atomic radius trends is not merely theoretical; it has significant practical applications.
11.1. Material Science
In material science, knowing the atomic radii helps in designing alloys with specific properties. For example, incorporating smaller atoms into a metal lattice can increase its strength.
11.2. Drug Design
In drug design, the size and shape of molecules, which are directly related to atomic radii, influence how drugs interact with biological targets.
11.3. Catalysis
In catalysis, atomic radii influence the surface area and reactivity of catalysts, affecting reaction rates and selectivity.
12. Recapping Key Concepts
Atomic radius is a critical concept in chemistry with periodic trends influenced by nuclear charge, shielding, and quantum numbers.
12.1. Key Takeaways
Atomic radii decrease across a period and increase down a group. Cations are smaller, and anions are larger than their parent atoms. Isoelectronic series show decreasing size with increasing nuclear charge.
12.2. Significance of Understanding Atomic Radius
Understanding atomic radius is essential for predicting chemical properties, designing materials, and developing new technologies.
13. Answering Frequently Asked Questions (FAQs)
Addressing common questions about atomic radius clarifies understanding.
13.1. What is atomic radius?
Atomic radius refers to the typical distance from the center of the nucleus to the boundary of the surrounding cloud of electrons.
13.2. How is atomic radius measured?
Atomic radius is typically measured using techniques like X-ray diffraction or quantum mechanical calculations, as direct measurement is not feasible.
13.3. Why does atomic radius decrease across a period?
Atomic radius decreases across a period due to increasing nuclear charge, which pulls electrons closer to the nucleus.
13.4. Why does atomic radius increase down a group?
Atomic radius increases down a group because electrons occupy larger orbitals with higher principal quantum numbers.
13.5. What is the difference between atomic and ionic radius?
Atomic radius refers to the size of a neutral atom, while ionic radius refers to the size of an ion (an atom that has gained or lost electrons).
13.6. Are cations larger or smaller than their parent atoms?
Cations are always smaller than their parent atoms because they have lost electrons, reducing electron-electron repulsion and increasing effective nuclear charge.
13.7. Are anions larger or smaller than their parent atoms?
Anions are always larger than their parent atoms because they have gained electrons, increasing electron-electron repulsion and decreasing effective nuclear charge.
13.8. What is an isoelectronic series?
An isoelectronic series is a group of ions or atoms that have the same number of electrons but different nuclear charges.
13.9. How does nuclear charge affect the size of ions in an isoelectronic series?
In an isoelectronic series, ions with a higher nuclear charge are smaller because the increased positive charge pulls the electrons closer to the nucleus.
13.10. What are some practical applications of understanding atomic radius?
Understanding atomic radius is crucial in materials science for designing alloys, in drug design for understanding molecular interactions, and in catalysis for optimizing reaction rates.
14. Final Thoughts and Recommendations
Mastering atomic radius concepts enhances your understanding of chemistry.
14.1. Importance of Periodic Trends
Understanding periodic trends is fundamental to predicting chemical behavior and designing new materials.
14.2. Further Resources on COMPARE.EDU.VN
COMPARE.EDU.VN offers detailed comparisons and insights into various scientific concepts, helping you make informed decisions and deepen your knowledge.
15. Call to Action
Navigating the complexities of comparing atomic radius can be challenging. At COMPARE.EDU.VN, we provide detailed and objective comparisons to help you make informed decisions. Whether you’re a student, a consumer, or a professional, our resources are designed to simplify the decision-making process.
Don’t struggle with complex comparisons alone. Visit COMPARE.EDU.VN today to explore comprehensive analyses and discover the best choices for your needs. Make your decisions with confidence, knowing you have the full picture at your fingertips.
For more information and assistance, contact us at:
- Address: 333 Comparison Plaza, Choice City, CA 90210, United States
- WhatsApp: +1 (626) 555-9090
- Website: COMPARE.EDU.VN
With compare.edu.vn, make the smart choice, every time.

