Heavy water, also known as deuterium oxide, exhibits unique properties when its behavior is compared to ordinary water, which this COMPARE.EDU.VN article will explore. Understanding these differences is crucial due to heavy water’s specific applications, especially in nuclear reactors. Explore the contrasting characteristics and applications of heavy water versus regular water, providing you with the knowledge to understand its significance.
1. Decoding Heavy Water: An Isotopic Dive
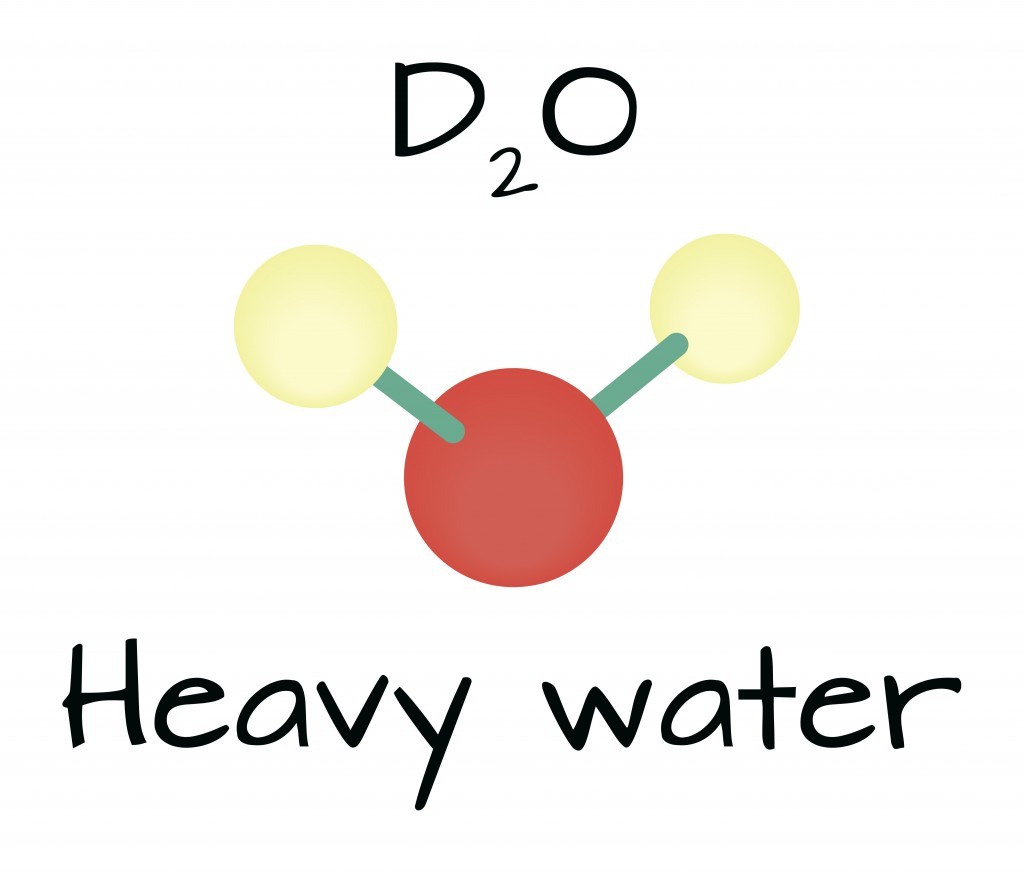
To truly understand the nuances of how heavy water behaves compared to regular water, it’s important to first understand its structure. Heavy water, or deuterium oxide (D2O), shares the same chemical structure as regular water (H2O) but differs in its isotopic composition. This seemingly small change leads to significant differences in physical and chemical properties.
1.1. Isotopes: The Foundation of Difference
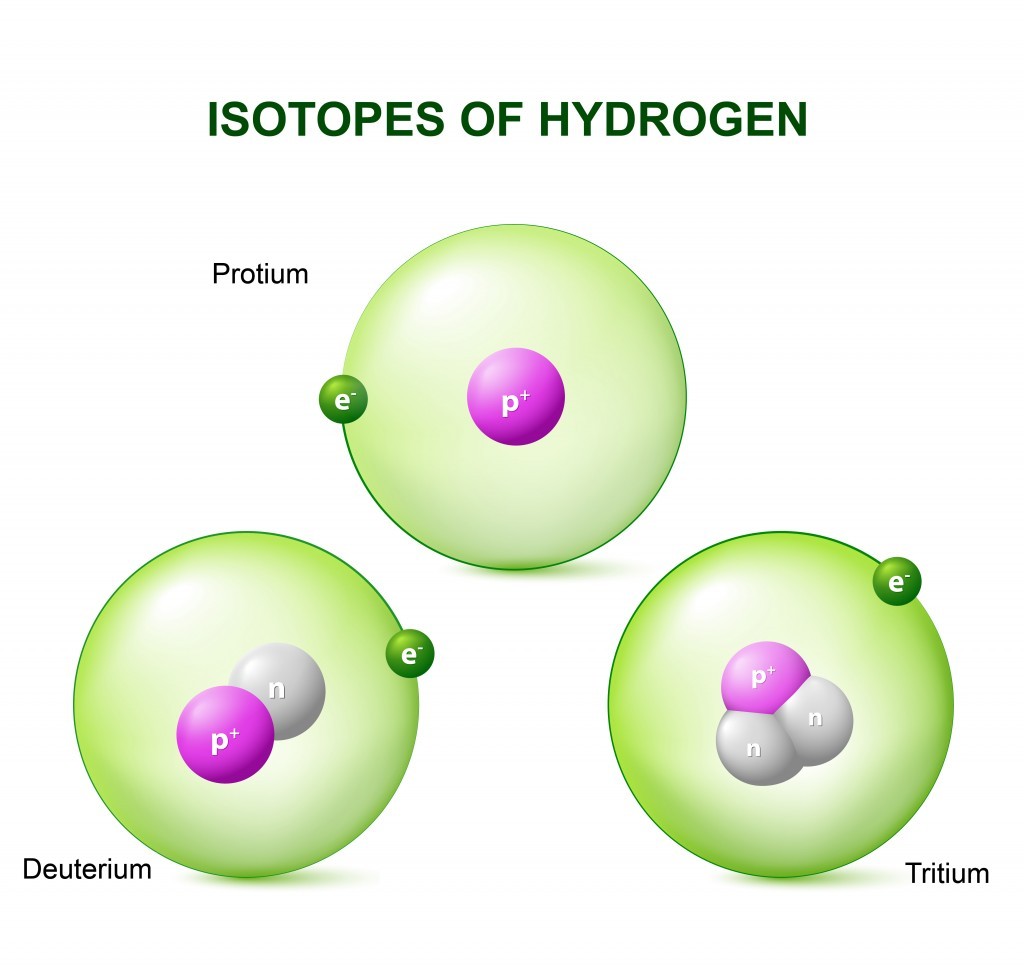
Isotopes are variants of a chemical element which differ in neutron number, and consequently in nucleon number. All isotopes of a given element have the same number of protons but different numbers of neutrons in each atom. Hydrogen, the key component of water, has three primary isotopes:
- Protium (¹H): The most common isotope, with one proton and no neutrons.
- Deuterium (²H or D): Contains one proton and one neutron, making it twice as heavy as protium.
- Tritium (³H or T): Contains one proton and two neutrons, making it three times heavier than protium and radioactive.
Alt: Diagram illustrating protium, deuterium, and tritium isotopes of hydrogen, highlighting differences in neutron count.
1.2. The Composition of Heavy Water
Regular water (H2O) consists of two protium atoms bonded to one oxygen atom. Heavy water (D2O), on the other hand, consists of two deuterium atoms bonded to one oxygen atom. This substitution of protium with deuterium is the primary factor influencing the differences in behavior between heavy water and regular water.
1.3. Molecular Weight and Density
The presence of deuterium significantly increases the molecular weight of heavy water. The molecular weight of H2O is approximately 18 atomic mass units (amu), while that of D2O is approximately 20 amu. This increase in molecular weight directly affects the density of heavy water. At room temperature, heavy water is about 11% more dense than regular water. This difference in density has several observable effects:
- Sinking Ice: Unlike regular ice, which floats on water, heavy ice (frozen D2O) sinks in regular water.
- Buoyancy: Objects that float easily in regular water may sink in heavy water due to the increased density.
2. Physical Properties: A Detailed Comparison
The physical properties of heavy water compared to regular water differ in several key aspects. These differences stem from the increased mass of deuterium, which affects the strength of hydrogen bonds and the overall behavior of the molecules.
2.1. Melting and Boiling Points
Heavy water has slightly different melting and boiling points compared to regular water:
- Melting Point: Regular water melts at 0°C (32°F), while heavy water melts at 3.82°C (38.88°F).
- Boiling Point: Regular water boils at 100°C (212°F), while heavy water boils at 101.42°C (214.56°F).
These differences are due to the stronger hydrogen bonds in heavy water, which require more energy to break, thus raising the melting and boiling points.
2.2. Density and Viscosity
As previously mentioned, heavy water is denser than regular water. This higher density affects its viscosity, which is a measure of a fluid’s resistance to flow:
- Density: Heavy water has a density of approximately 1.107 g/cm³ at 20°C, compared to regular water’s density of 0.998 g/cm³.
- Viscosity: Heavy water is more viscous than regular water. This means it flows more slowly. At 25°C, the viscosity of heavy water is about 25% greater than that of regular water.
These differences in density and viscosity can affect various processes, from chemical reactions to biological functions.
2.3. Hydrogen Bonding
Hydrogen bonds are crucial in determining the properties of water. They are formed between the slightly positive hydrogen atom of one water molecule and the slightly negative oxygen atom of another. In heavy water, these bonds are slightly stronger due to the increased mass of deuterium:
- Bond Strength: The stronger hydrogen bonds in heavy water require more energy to break, leading to higher melting and boiling points.
- Molecular Arrangement: The stronger bonds also influence the arrangement of molecules in the liquid state, affecting its structure and behavior.
2.4. Spectroscopic Properties
Heavy water exhibits different spectroscopic properties compared to regular water. Spectroscopy involves studying the interaction of electromagnetic radiation with matter. The differences arise because deuterium’s heavier mass affects the vibrational and rotational frequencies of the water molecule:
- Infrared (IR) Spectroscopy: Heavy water absorbs infrared radiation at different frequencies than regular water, allowing scientists to distinguish between the two using IR spectroscopy.
- Nuclear Magnetic Resonance (NMR) Spectroscopy: Deuterium has different NMR properties than protium, which allows for the identification and quantification of heavy water in a sample.
These spectroscopic differences are valuable in scientific research and industrial applications for analyzing and monitoring heavy water.
3. Chemical Properties: How Reactions Differ
The chemical properties of heavy water compared to regular water also exhibit notable differences. These variations primarily arise from the kinetic isotope effect (KIE), which influences reaction rates and equilibrium constants.
3.1. Kinetic Isotope Effect (KIE)
The kinetic isotope effect (KIE) is the change in the rate of a chemical reaction when one of the atoms in the reactants is replaced by one of its isotopes. In the case of heavy water, the substitution of protium with deuterium affects the reaction rate:
- Reaction Rates: Reactions involving heavy water tend to be slower than those involving regular water. This is because the heavier deuterium atom forms stronger bonds, requiring more energy to break.
- Primary KIE: Occurs when the bond to the isotopically substituted atom is broken in the rate-determining step of the reaction.
- Secondary KIE: Occurs when the isotopic substitution is not directly involved in bond breaking but still affects the reaction rate due to changes in vibrational frequencies.
3.2. Acidity and Basicity
Heavy water is slightly less acidic and less basic than regular water:
- Acidity: The pD (analogous to pH for heavy water) of neutral heavy water is 7.41 at 25°C, compared to a pH of 7.0 for regular water. This means heavy water is slightly less acidic.
- Basicity: Similarly, heavy water is slightly less basic than regular water.
These differences in acidity and basicity can affect the outcome of acid-base catalyzed reactions.
3.3. Solubility
The solubility of certain compounds can vary in heavy water compared to regular water:
- Ionic Compounds: Some ionic compounds may exhibit slightly different solubilities in heavy water due to the differences in ion-solvent interactions.
- Organic Compounds: Organic compounds may also show variations in solubility, particularly those that can form hydrogen bonds with the solvent.
3.4. Biochemical Reactions
In biological systems, heavy water can significantly affect biochemical reactions:
- Enzyme Activity: Enzymes may exhibit altered activity in heavy water due to changes in the rate of proton transfer reactions.
- DNA and Protein Stability: Heavy water can influence the stability of DNA and proteins, affecting their structure and function.
Due to these effects, heavy water is often used in biochemical research to study reaction mechanisms and protein dynamics.
Alt: Molecular structure of heavy water (D2O), highlighting deuterium atoms.
4. Applications of Heavy Water
Heavy water’s unique properties make it valuable in various scientific, industrial, and research applications.
4.1. Nuclear Reactors
One of the primary uses of heavy water is in nuclear reactors:
- Neutron Moderator: Heavy water is an excellent neutron moderator, slowing down neutrons to sustain the nuclear chain reaction in reactors using natural uranium as fuel.
- CANDU Reactors: The CANDU (Canadian Deuterium Uranium) reactor design uses heavy water as both a moderator and a coolant.
4.2. Nuclear Magnetic Resonance (NMR) Spectroscopy
Heavy water is widely used as a solvent in NMR spectroscopy:
- Deuterium Lock: Deuterium provides a stable signal for field-frequency locking in NMR spectrometers.
- Signal Simplification: Replacing protium with deuterium simplifies NMR spectra by eliminating protium signals from the solvent.
4.3. Isotopic Tracers
Heavy water can be used as an isotopic tracer in various studies:
- Metabolic Studies: Deuterated compounds can be used to trace metabolic pathways in organisms.
- Hydrological Studies: Heavy water can be used to study water movement and distribution in hydrological systems.
4.4. Research in Chemistry and Biology
Heavy water is an essential tool in chemical and biological research:
- Reaction Mechanisms: Studying reactions in heavy water helps elucidate reaction mechanisms and transition states.
- Protein Dynamics: Heavy water is used to investigate protein folding, stability, and interactions.
5. Safety and Biological Effects
While heavy water is not radioactive, it can have biological effects if consumed in large quantities.
5.1. Toxicity
- Small Quantities: Small amounts of heavy water are not harmful. The human body naturally contains trace amounts of deuterium.
- Large Quantities: Consuming large amounts of heavy water can disrupt normal physiological processes. If 20% or more of the water in the body is replaced by heavy water, it can lead to adverse health effects.
5.2. Physiological Effects
- Cellular Function: Heavy water can slow down cellular processes and enzyme activity.
- Mammalian Studies: Studies on mammals have shown that high concentrations of heavy water can lead to sterility, reduced growth rates, and other health problems.
5.3. Precautions
- Handling: While not highly toxic, heavy water should be handled with care, especially in laboratory settings.
- Consumption: Avoid consuming large quantities of heavy water.
Alt: Illustration of a toxic hazard symbol, cautioning against the consumption of excess heavy water.
6. Detailed Comparison Table: Heavy Water vs. Regular Water
| Property | Regular Water (H2O) | Heavy Water (D2O) | Difference |
|---|---|---|---|
| Molecular Weight | 18.015 amu | 20.027 amu | Heavier by ~11% |
| Melting Point | 0°C (32°F) | 3.82°C (38.88°F) | Higher by 3.82°C |
| Boiling Point | 100°C (212°F) | 101.42°C (214.56°F) | Higher by 1.42°C |
| Density (at 20°C) | 0.998 g/cm³ | 1.107 g/cm³ | Denser by ~11% |
| Viscosity (at 25°C) | 0.890 cP | 1.10 cP | More viscous by ~25% |
| Acidity (pKa) | 14.00 | 14.87 | Less acidic |
| Basicity (pKb) | 14.00 | 14.87 | Less basic |
| Neutron Moderation | Poor | Excellent | Significantly better neutron moderation |
| Biological Effects | Essential for life | Toxic in large doses | Can disrupt biological processes |
7. The Economic and Production Aspects of Heavy Water
7.1. Production Methods
Heavy water production is an intricate process. The most common method is the Girdler sulfide process or GS process. The GS process relies on the difference in the equilibrium constant between hydrogen sulfide and water at different temperatures. This chemical exchange is carried out in a series of towers.
7.2. Cost of Heavy Water
The cost of heavy water is considerable, reflecting the energy-intensive processes required for its production. As of recent estimates, heavy water can cost hundreds of dollars per kilogram. This high cost influences its use, often limiting it to applications where its unique properties are critical.
7.3. Global Producers
Countries with substantial nuclear programs are often the primary producers of heavy water. Canada, India, Argentina, and Russia have significant heavy water production capabilities.
8. Environmental Considerations
8.1. Potential Contamination
Although heavy water is not radioactive, its release into the environment can have ecological consequences. High concentrations of heavy water can negatively impact aquatic life and disrupt ecosystems.
8.2. Regulatory Oversight
Stringent regulations oversee the production, handling, and disposal of heavy water to prevent environmental contamination and ensure safety.
9. Heavy Water in Popular Culture and Media
9.1. Misconceptions
Despite its importance in scientific and industrial applications, heavy water is often misunderstood in popular culture. It is sometimes mistakenly associated with radioactivity or perceived as a dangerous substance.
9.2. Fictional Depictions
Heavy water has appeared in various works of fiction, often as a critical component in nuclear-related storylines. These depictions sometimes exaggerate its properties or dangers, contributing to public misconceptions.
10. Future Trends and Research
10.1. Advanced Reactor Designs
Ongoing research focuses on advanced nuclear reactor designs that may utilize heavy water more efficiently or explore alternative moderators.
10.2. Biomedical Applications
Continued studies investigate the potential biomedical applications of deuterium and heavy water. Research areas include drug development, metabolic studies, and diagnostic imaging.
10.3. Isotope Separation Technologies
Advances in isotope separation technologies could lead to more efficient and cost-effective heavy water production methods.
11. Addressing Common Misconceptions About Heavy Water
11.1. Is Heavy Water Radioactive?
One of the most pervasive myths about heavy water is that it is radioactive. This misconception likely arises from its association with nuclear reactors and nuclear energy.
11.2. Can You Drink Heavy Water?
While technically possible to consume heavy water, the question is whether it’s safe and advisable. Consuming small amounts of heavy water is not immediately harmful because our bodies naturally contain trace amounts of deuterium.
11.3. Does Heavy Water Exist in Nature?
Many people are surprised to learn that heavy water is not a purely synthetic substance created in laboratories. Heavy water exists naturally on Earth, though in very small concentrations.
12. Heavy Water and the Future of Nuclear Energy
12.1. Efficiency in Nuclear Reactors
One of the key advantages of using heavy water in nuclear reactors is its efficiency as a neutron moderator. Neutron moderators are substances used in nuclear reactors to slow down neutrons, increasing the probability of nuclear fission.
12.2. Implications for Sustainable Energy
As the world increasingly seeks sustainable energy solutions, the role of nuclear energy, particularly with heavy water reactors, becomes more significant. Heavy water reactors, especially CANDU reactors, can operate using natural uranium.
12.3. Safety Measures and Technological Advances
Continuous advancements in reactor technology and safety measures have made heavy water reactors safer and more efficient. These include enhanced monitoring systems, improved containment structures, and advanced control mechanisms.
13. Distinguishing Heavy Water from Tritiated Water
13.1. Tritiated Water: An Overview
Tritiated water is water in which one or both of the hydrogen atoms are replaced with tritium, a radioactive isotope of hydrogen. Understanding the properties and implications of tritiated water is essential to fully grasp the scope of heavy water and its counterparts.
13.2. Environmental Concerns
The presence of tritium in water poses significant environmental concerns. Tritium is a radioactive isotope, and its release into the environment can lead to contamination.
13.3. Contrasting Safety Profiles
Heavy water and tritiated water have vastly different safety profiles. Heavy water, while capable of causing physiological disruptions in high concentrations, is not radioactive.
14. Exploring the Applications of Deuterium in Scientific Research
14.1. NMR Spectroscopy
Nuclear Magnetic Resonance (NMR) spectroscopy is a powerful technique used to study the structure and dynamics of molecules. Deuterium plays a crucial role in NMR spectroscopy.
14.2. Chemical Kinetics
Deuterium is extensively used in chemical kinetics to study reaction mechanisms. Replacing hydrogen with deuterium can alter the rate of a chemical reaction.
14.3. Materials Science
In materials science, deuterium is used to modify the properties of various materials. For example, deuterium implantation can alter the surface characteristics of semiconductors.
15. Heavy Water: A Comparative Look at Production Costs
15.1. Capital Investment
The establishment of heavy water production facilities requires substantial capital investment. The construction of GS process plants, for example, involves the creation of large-scale industrial infrastructure.
15.2. Energy Consumption
Heavy water production is an energy-intensive process. The GS process, in particular, requires significant amounts of energy to maintain the necessary temperature and pressure conditions.
15.3. Operational Expenses
Ongoing operational expenses contribute to the overall cost of heavy water production. These expenses include the cost of raw materials.
16. The Future Outlook: Innovations in Heavy Water Production
16.1. Advanced Separation Techniques
Researchers are continually exploring advanced separation techniques to improve the efficiency of heavy water production. These techniques include laser-based separation methods and advanced chemical exchange processes.
16.2. Sustainable Production Methods
The development of more sustainable heavy water production methods is a growing area of interest. This includes exploring the use of renewable energy sources to power production facilities.
16.3. Economic Viability
Improving the economic viability of heavy water production is essential for its continued use in various applications. Efforts are focused on reducing production costs, improving energy efficiency.
17. FAQ: Addressing Common Queries About Heavy Water
1. Is heavy water radioactive?
No, heavy water (D2O) is not radioactive. It contains deuterium, a stable isotope of hydrogen, not a radioactive one.
2. Can I drink heavy water?
Small amounts are not harmful, but consuming large quantities can disrupt bodily functions.
3. What is heavy water used for?
It’s primarily used in nuclear reactors as a neutron moderator and in NMR spectroscopy.
4. How does heavy water differ from regular water?
Heavy water is denser, has slightly higher melting and boiling points, and affects reaction rates differently.
5. Does heavy water occur naturally?
Yes, but in very small concentrations compared to regular water.
6. Is heavy water toxic?
In large doses, it can be toxic due to its effect on biochemical reactions.
7. What is the kinetic isotope effect?
It’s the change in the rate of a chemical reaction when an atom is replaced by its isotope, like hydrogen by deuterium.
8. How is heavy water produced?
The most common method is the Girdler sulfide (GS) process.
9. What are the environmental concerns related to heavy water?
Although it is not radioactive, its release into the environment in high concentrations can disrupt ecosystems.
10. Is tritiated water the same as heavy water?
No, tritiated water contains tritium, a radioactive isotope of hydrogen, while heavy water contains deuterium, a stable isotope.
18. Conclusion: Making Informed Decisions with COMPARE.EDU.VN
Understanding how heavy water behaves compared to regular water is crucial for various applications, from nuclear energy to scientific research. The differences in physical and chemical properties, as well as the safety considerations, make it essential to approach heavy water with informed awareness. Navigate the complexities of product comparisons with ease by visiting COMPARE.EDU.VN. Our comprehensive comparison tools empower you to make confident decisions.
Ready to make smarter choices? Visit COMPARE.EDU.VN today and explore detailed comparisons across various products and services. Our platform offers in-depth analysis and user reviews to help you find the best options tailored to your needs. Don’t settle for less—discover the COMPARE.EDU.VN difference!
Address: 333 Comparison Plaza, Choice City, CA 90210, United States
WhatsApp: +1 (626) 555-9090
Website: compare.edu.vn