How Do The Results Of The Pour Plate Method Compare? The pour plate method is a valuable technique in microbiology for quantifying and isolating microorganisms. At COMPARE.EDU.VN, we provide comprehensive comparisons to help you understand its advantages and limitations compared to other methods, assisting you in making informed decisions in your scientific endeavors. Explore detailed breakdowns and expert analysis that offer a clear understanding of microbial enumeration techniques.
1. Understanding Microbial Enumeration Techniques
Microbial enumeration techniques are crucial for assessing the quantity of microorganisms in various samples. Different methods offer distinct advantages and disadvantages. The key is selecting the most appropriate method for your specific application. This section provides an overview of several common techniques, including streak plating, pour plating, and spread plating.
1.1 Streak Plate Technique
The streak plate technique is a qualitative method used to isolate pure cultures of bacteria from a mixed population. A loop is used to spread the sample across the agar surface, gradually diluting the cells to obtain single colonies.
- Application: Isolating single colonies from mixed cultures.
- Procedure: A sterile loop is used to streak a sample across the agar surface in a quadrant pattern, gradually diluting the sample to obtain isolated colonies.
- Advantages: Simple, inexpensive, and effective for obtaining pure cultures.
- Disadvantages: Not suitable for quantifying bacterial populations, requires skill to perform effectively.
1.2 Pour Plate Technique
The pour plate technique is a quantitative method used to determine the number of viable cells in a sample. The sample is mixed with molten agar, which is then poured into a sterile Petri dish and allowed to solidify. Colonies form both on the surface and within the agar.
- Application: Determining the number of viable cells in a sample.
- Procedure: A sample is mixed with molten agar, poured into a Petri dish, and allowed to solidify. Colonies form both on the surface and within the agar.
- Advantages: Can handle larger sample volumes, colonies are distributed throughout the agar.
- Disadvantages: Heat from the molten agar may harm some microorganisms, colonies within the agar may be difficult to count, anaerobic conditions may develop within the agar.
1.3 Spread Plate Technique
The spread plate technique is another quantitative method used for viable cell counts. A small volume of sample is spread evenly over the surface of an agar plate. Colonies form only on the surface of the agar.
- Application: Determining the number of viable cells in a sample, isolating single colonies.
- Procedure: A small volume of sample is spread evenly over the surface of an agar plate using a sterile spreader. Colonies form only on the surface of the agar.
- Advantages: Colonies are easily accessible for further study, no heat exposure.
- Disadvantages: Can only handle small sample volumes, requires more skill to ensure even spreading.
1.4 Soft Agar Overlay Technique
The soft agar overlay technique is used to count bacteriophages by mixing the sample with host cells in soft agar, which is then overlaid on a hard agar plate. Plaques (zones of clearing) form where the phages have infected and lysed the host cells.
- Application: Counting bacteriophages.
- Procedure: Phage particles are mixed with host cells in soft agar, overlaid on a hard agar plate, and incubated. Plaques form where phages lyse host cells.
- Advantages: Allows for easy visualization and counting of phage plaques.
- Disadvantages: Requires specific host cells, technique-sensitive.
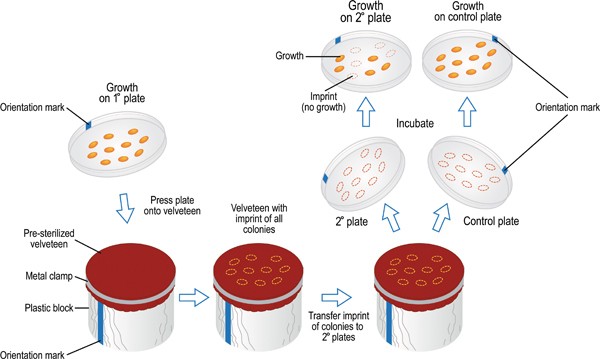
1.5 Replica Plating
Replica plating is a technique used to simultaneously screen a large number of microorganisms for specific growth requirements. Colonies from a master plate are transferred to multiple secondary plates with different media.
- Application: Screening microorganisms for specific growth requirements.
- Procedure: Colonies from a master plate are transferred to multiple secondary plates with different media using a sterile velveteen cloth.
- Advantages: Allows for rapid screening of many colonies under different conditions.
- Disadvantages: Requires careful technique to avoid contamination, may produce false positives due to nutrient carryover.
2. Pour Plate Method: A Deep Dive
The pour plate method is a versatile technique that provides valuable insights into the microbial composition of a sample. This section explores the principles, applications, advantages, and disadvantages of the pour plate method in detail.
2.1 Principle of the Pour Plate Method
The pour plate method involves mixing a sample containing microorganisms with molten agar at a temperature that will not kill the microorganisms (typically around 45-50°C). This mixture is then poured into a sterile Petri dish and allowed to solidify. As the agar solidifies, the microorganisms are evenly distributed throughout the medium.
2.2 Applications of the Pour Plate Method
The pour plate method has numerous applications across various fields, making it an indispensable tool for microbial enumeration and analysis.
- Water Quality Testing: Assessing the microbial load in water samples to ensure safety for drinking and other uses.
- Food Microbiology: Evaluating the microbial content of food products to determine spoilage and safety.
- Pharmaceutical Microbiology: Monitoring the microbial contamination levels in pharmaceutical products during production and storage.
- Environmental Microbiology: Analyzing soil and water samples to study microbial communities.
- Clinical Microbiology: Enumerating bacteria in clinical samples for diagnostic purposes.
2.3 Advantages of the Pour Plate Method
The pour plate method offers several advantages that make it a preferred choice for certain applications.
- Large Sample Volume: Can accommodate larger sample volumes compared to the spread plate method.
- Colony Distribution: Colonies are distributed throughout the agar, allowing for better representation of the sample.
- Quantitative Analysis: Provides a reliable estimate of the number of viable cells in a sample.
- Versatility: Suitable for a wide range of microorganisms and sample types.
2.4 Disadvantages of the Pour Plate Method
Despite its advantages, the pour plate method also has certain limitations.
- Heat Sensitivity: The heat from the molten agar can kill or inhibit the growth of some heat-sensitive microorganisms.
- Anaerobic Conditions: Colonies embedded within the agar may experience anaerobic conditions, which can affect their growth.
- Counting Difficulties: Colonies within the agar can be difficult to count compared to those on the surface.
- Colony Morphology: The morphology of colonies within the agar may differ from those on the surface, making identification challenging.
3. How Do the Results of the Pour Plate Method Compare to Other Techniques?
To fully appreciate the strengths and weaknesses of the pour plate method, it is essential to compare its results with those obtained from other microbial enumeration techniques. This section provides a detailed comparison of the pour plate method with the streak plate, spread plate, soft agar overlay, and replica plating techniques.
3.1 Pour Plate vs. Streak Plate
The streak plate method is primarily used for isolating pure cultures rather than quantifying microbial populations. In contrast, the pour plate method provides a quantitative estimate of the number of viable cells in a sample.
| Feature | Pour Plate | Streak Plate |
|---|---|---|
| Primary Use | Quantitative enumeration of viable cells | Isolation of pure cultures |
| Colony Location | Surface and within the agar | Surface only |
| Sample Volume | Can handle larger volumes | Limited to small volumes |
| Heat Exposure | Microorganisms exposed to molten agar heat | No heat exposure |
| Quantitative | Yes | No |
| Skill Level | Moderate | Requires practice to master |
| Aerobic/Anaerobic | Anaerobic conditions possible within agar | Aerobic conditions |
3.2 Pour Plate vs. Spread Plate
Both the pour plate and spread plate methods are used for quantitative enumeration of microorganisms, but they differ in how the sample is applied to the agar. The spread plate method avoids heat exposure, while the pour plate method can handle larger sample volumes.
| Feature | Pour Plate | Spread Plate |
|---|---|---|
| Primary Use | Quantitative enumeration of viable cells | Quantitative enumeration of viable cells |
| Colony Location | Surface and within the agar | Surface only |
| Sample Volume | Can handle larger volumes | Limited to small volumes |
| Heat Exposure | Microorganisms exposed to molten agar heat | No heat exposure |
| Quantitative | Yes | Yes |
| Skill Level | Moderate | Requires skill to spread evenly |
| Aerobic/Anaerobic | Anaerobic conditions possible within agar | Aerobic conditions |
3.3 Pour Plate vs. Soft Agar Overlay
The soft agar overlay technique is mainly used for counting bacteriophages, whereas the pour plate method is used for enumerating bacteria and other microorganisms.
| Feature | Pour Plate | Soft Agar Overlay |
|---|---|---|
| Primary Use | Quantitative enumeration of viable cells | Counting bacteriophages |
| Colony Location | Surface and within the agar | Plaques on a bacterial lawn |
| Sample Volume | Can handle larger volumes | Specialized for phage assays |
| Heat Exposure | Microorganisms exposed to molten agar heat | Soft agar cooled before mixing with phage |
| Quantitative | Yes | Yes |
| Skill Level | Moderate | Requires specific expertise |
| Aerobic/Anaerobic | Anaerobic conditions possible within agar | Aerobic conditions |
3.4 Pour Plate vs. Replica Plating
Replica plating is used for screening microorganisms for specific growth requirements, while the pour plate method is used for quantitative enumeration.
| Feature | Pour Plate | Replica Plating |
|---|---|---|
| Primary Use | Quantitative enumeration of viable cells | Screening for growth requirements |
| Colony Location | Surface and within the agar | Surface only |
| Sample Volume | Can handle larger volumes | Not applicable |
| Heat Exposure | Microorganisms exposed to molten agar heat | No heat exposure |
| Quantitative | Yes | No |
| Skill Level | Moderate | Requires careful technique |
| Aerobic/Anaerobic | Anaerobic conditions possible within agar | Aerobic conditions |
4. Factors Affecting Pour Plate Results
Several factors can influence the results obtained from the pour plate method. Understanding these factors is crucial for ensuring accurate and reliable data.
4.1 Temperature of Molten Agar
The temperature of the molten agar is a critical factor. If the agar is too hot (above 50°C), it can kill or inhibit the growth of heat-sensitive microorganisms, leading to an underestimation of the viable cell count. Conversely, if the agar is too cool, it may begin to solidify before being poured, resulting in uneven distribution of microorganisms.
4.2 Mixing Technique
Proper mixing of the sample with the molten agar is essential for even distribution of microorganisms. Insufficient mixing can lead to clumping of cells, resulting in inaccurate colony counts. Gentle swirling is recommended to ensure uniform distribution without introducing air bubbles.
4.3 Incubation Conditions
Incubation temperature, duration, and atmospheric conditions can significantly affect colony growth. Different microorganisms have different optimal growth conditions. It is important to incubate the plates under the appropriate conditions to ensure optimal growth and accurate enumeration.
4.4 Sample Preparation
The accuracy of pour plate results depends on proper sample preparation. The sample should be properly diluted to obtain a countable number of colonies (typically between 30 and 300 colonies per plate). Serial dilutions are often necessary to achieve the appropriate concentration.
4.5 Media Composition
The composition of the agar medium can also affect colony growth. The medium should provide all the necessary nutrients for the microorganisms being enumerated. Selective media can be used to inhibit the growth of unwanted microorganisms and promote the growth of specific target organisms.
5. Troubleshooting Common Issues in Pour Plate Method
Even with careful technique, issues can arise during the pour plate method. This section provides troubleshooting tips for some common problems.
5.1 No Colony Growth
- Problem: No colonies appear on the plates.
- Possible Causes:
- Agar too hot, killing microorganisms.
- Inhibitory substances in the medium.
- Incorrect incubation conditions.
- Sample not viable.
- Solutions:
- Ensure agar temperature is appropriate (45-50°C).
- Check the medium for inhibitory substances.
- Verify incubation conditions are correct.
- Confirm sample viability.
5.2 Too Many Colonies
- Problem: Plates are overcrowded with colonies, making counting impossible.
- Possible Causes:
- Insufficient dilution of the sample.
- Contamination.
- Solutions:
- Increase the dilution factor.
- Ensure sterile technique.
- Prepare fresh media.
5.3 Uneven Colony Distribution
- Problem: Colonies are not evenly distributed across the agar.
- Possible Causes:
- Inadequate mixing of the sample with the molten agar.
- Agar solidifying too quickly.
- Solutions:
- Ensure thorough mixing of the sample and agar.
- Work quickly to pour the plates before the agar solidifies.
5.4 Colonies Only on the Surface
- Problem: Colonies only grow on the surface of the agar.
- Possible Causes:
- Agar poured too hot, killing subsurface organisms.
- Microorganisms are strict aerobes.
- Solutions:
- Ensure agar temperature is appropriate.
- Consider using anaerobic incubation for anaerobic organisms.
5.5 Contamination
- Problem: Unwanted microorganisms appear on the plates.
- Possible Causes:
- Non-sterile equipment or media.
- Airborne contamination.
- Solutions:
- Ensure all equipment and media are sterile.
- Work in a clean environment (e.g., laminar flow hood).
- Use proper aseptic techniques.
6. Pour Plate Method in Industrial and Environmental Applications
The pour plate method is widely used in industrial and environmental settings to monitor and control microbial populations. This section explores some specific applications in these fields.
6.1 Water Quality Monitoring
The pour plate method is a standard technique for assessing the microbial quality of water. It is used to enumerate total bacteria, coliforms, and other indicator organisms in drinking water, recreational water, and wastewater. Regulatory agencies often specify the pour plate method for compliance monitoring.
- Application: Determining the safety of drinking water by counting total bacteria and coliforms.
- Procedure: Water samples are serially diluted and mixed with molten agar. After incubation, colonies are counted, and the number of bacteria per milliliter is calculated. Selective media can be used to enumerate specific types of bacteria.
- Significance: Ensures water is safe for consumption and meets regulatory standards.
6.2 Food Safety
In the food industry, the pour plate method is used to monitor the microbial content of food products to ensure safety and prevent spoilage. It is used to enumerate bacteria, yeast, and mold in raw materials, in-process samples, and finished products.
- Application: Monitoring the microbial content of food products to prevent spoilage and ensure safety.
- Procedure: Food samples are homogenized, serially diluted, and mixed with molten agar. After incubation, colonies are counted, and the number of microorganisms per gram is calculated.
- Significance: Prevents foodborne illnesses and ensures product quality.
6.3 Pharmaceutical Quality Control
Pharmaceutical companies use the pour plate method to monitor the microbial contamination of pharmaceutical products, manufacturing environments, and equipment. This helps to ensure the sterility and safety of drugs and medical devices.
- Application: Monitoring the microbial contamination of pharmaceutical products and manufacturing environments.
- Procedure: Samples from pharmaceutical products or manufacturing surfaces are mixed with molten agar. After incubation, colonies are counted, and the level of contamination is determined.
- Significance: Ensures the sterility and safety of pharmaceutical products.
6.4 Environmental Monitoring
The pour plate method is used in environmental monitoring to assess the microbial quality of soil, sediments, and air. It is used to enumerate bacteria, fungi, and other microorganisms in environmental samples.
- Application: Assessing the microbial quality of soil, sediments, and air samples.
- Procedure: Environmental samples are processed, serially diluted, and mixed with molten agar. After incubation, colonies are counted, and the number of microorganisms per gram or liter is calculated.
- Significance: Provides insights into the health and function of environmental ecosystems.
7. Optimizing Pour Plate Results: Best Practices
To achieve accurate and reliable results with the pour plate method, it is important to follow best practices in technique, materials, and data analysis. This section outlines key recommendations for optimizing pour plate results.
7.1 Standardize Technique
- Consistent Pipetting: Use calibrated pipettes and practice consistent pipetting techniques to ensure accurate sample volumes.
- Temperature Control: Monitor the temperature of the molten agar using a thermometer to ensure it remains within the optimal range (45-50°C).
- Mixing Protocol: Use a standardized mixing protocol, such as gentle swirling, to ensure uniform distribution of microorganisms.
- Pouring Time: Minimize the time between mixing the sample with the agar and pouring the plates to prevent premature solidification.
7.2 Use High-Quality Materials
- Sterile Media: Use commercially prepared sterile agar media or prepare media according to established protocols and sterilize by autoclaving.
- Sterile Equipment: Use sterile Petri dishes, pipettes, and other equipment to prevent contamination.
- Certified Reagents: Use certified reagents and chemicals to ensure the accuracy and reliability of the media.
7.3 Control Incubation Conditions
- Accurate Temperature: Use a calibrated incubator to maintain the correct incubation temperature.
- Humidity Control: Monitor humidity levels in the incubator to prevent dehydration of the agar.
- Atmospheric Control: Use anaerobic jars or incubators for anaerobic organisms.
- Consistent Incubation Time: Incubate plates for the recommended time to allow for optimal colony growth.
7.4 Proper Data Analysis
- Colony Counting: Use a colony counter or manual counting techniques to accurately enumerate colonies on the plates.
- CFU Calculation: Calculate colony-forming units (CFU) per milliliter or gram using the appropriate dilution factors.
- Statistical Analysis: Perform statistical analysis to assess the variability and significance of the results.
- Quality Control: Include positive and negative controls to validate the accuracy of the pour plate method.
8. Advantages of Using COMPARE.EDU.VN for Microbial Enumeration Insights
COMPARE.EDU.VN provides comprehensive and objective comparisons of microbial enumeration techniques, helping you make informed decisions for your specific needs. Our platform offers detailed analyses, expert opinions, and user reviews to guide you through the complexities of microbial quantification.
8.1 Comprehensive Comparisons
COMPARE.EDU.VN offers in-depth comparisons of the pour plate method with other microbial enumeration techniques, including streak plating, spread plating, soft agar overlay, and replica plating. Our comparisons cover the principles, applications, advantages, and disadvantages of each method, enabling you to select the most appropriate technique for your research or application.
8.2 Expert Analysis
Our team of experts provides detailed analyses of the factors affecting pour plate results, including temperature, mixing technique, incubation conditions, sample preparation, and media composition. We offer troubleshooting tips for common issues and best practices for optimizing your results.
8.3 Real-World Applications
COMPARE.EDU.VN showcases real-world applications of the pour plate method in various industries, including water quality monitoring, food safety, pharmaceutical quality control, and environmental monitoring. Our case studies and examples illustrate the versatility and importance of the pour plate method in practical settings.
8.4 User Reviews and Ratings
Our platform features user reviews and ratings of different pour plate techniques and materials, providing valuable insights from fellow researchers and practitioners. You can benefit from the experiences of others to improve your own pour plate procedures.
8.5 Easy-to-Use Interface
COMPARE.EDU.VN offers an intuitive and user-friendly interface, making it easy to navigate and find the information you need. Our search and filter functions allow you to quickly locate specific comparisons, analyses, and reviews.
9. Conclusion: Making Informed Decisions with COMPARE.EDU.VN
The pour plate method is a valuable tool for quantitative microbial enumeration, offering unique advantages for certain applications. By understanding its principles, applications, advantages, and limitations, you can make informed decisions about when and how to use this technique. COMPARE.EDU.VN provides the resources and insights you need to master the pour plate method and other microbial enumeration techniques. Whether you are a researcher, a student, or an industry professional, our platform is your trusted source for objective and comprehensive comparisons.
Ready to take your microbial enumeration skills to the next level? Visit COMPARE.EDU.VN today to explore our detailed comparisons and expert analyses. Make informed decisions and achieve accurate, reliable results with our comprehensive guidance.
For further information, contact us at:
Address: 333 Comparison Plaza, Choice City, CA 90210, United States
WhatsApp: +1 (626) 555-9090
Website: compare.edu.vn
10. Frequently Asked Questions (FAQs)
10.1 What is the main purpose of the pour plate method?
The primary purpose of the pour plate method is to quantitatively determine the number of viable microorganisms in a sample by mixing it with molten agar and counting the resulting colonies after incubation.
10.2 How does the pour plate method differ from the spread plate method?
In the pour plate method, the sample is mixed with molten agar before pouring it into a Petri dish, allowing colonies to grow both on the surface and within the agar. In the spread plate method, the sample is spread on the surface of an already solidified agar plate, resulting in colonies growing only on the surface.
10.3 What are the advantages of using the pour plate method?
The pour plate method can handle larger sample volumes, provides a more representative distribution of colonies, and is suitable for a wide range of microorganisms and sample types.
10.4 What are the disadvantages of using the pour plate method?
The heat from the molten agar can harm heat-sensitive microorganisms, colonies within the agar may experience anaerobic conditions, and counting colonies within the agar can be difficult.
10.5 How can I ensure accurate results with the pour plate method?
To ensure accurate results, standardize your technique, use high-quality materials, control incubation conditions, and perform proper data analysis.
10.6 What temperature should the molten agar be when using the pour plate method?
The molten agar should be between 45-50°C to avoid killing heat-sensitive microorganisms while ensuring it remains liquid enough to mix with the sample.
10.7 How do I prevent contamination when using the pour plate method?
Use sterile equipment and media, work in a clean environment (e.g., laminar flow hood), and employ proper aseptic techniques to prevent contamination.
10.8 Can the pour plate method be used for both aerobic and anaerobic bacteria?
Yes, but for anaerobic bacteria, you need to incubate the plates under anaerobic conditions using anaerobic jars or incubators.
10.9 What should I do if I see no colony growth on my pour plates?
Check the agar temperature, look for inhibitory substances in the medium, verify incubation conditions, and confirm sample viability.
10.10 How do I calculate colony-forming units (CFU) from pour plate results?
Count the number of colonies on a plate with between 30 and 300 colonies, and then calculate CFU per milliliter or gram using the appropriate dilution factors.