How Are Lipids Unique When Compared To Other Macromolecule Groups? Lipids, essential for life, boast unique characteristics setting them apart. COMPARE.EDU.VN offers comprehensive comparisons to understand these differences. Exploring their distinct properties unveils their critical roles in biology, health, and nutrition. Discover more about these vital biomolecules, lipid functions, and structural lipids only at COMPARE.EDU.VN.
1. Introduction to Macromolecules and Lipids
Biological macromolecules are large organic molecules crucial for life. They fall into four major categories: carbohydrates, lipids (or fats), proteins, and nucleic acids. Each group plays a distinct role in the structure and function of cells and organisms. Among these, lipids stand out due to their unique chemical properties and diverse functions. This article, presented by COMPARE.EDU.VN, will explore “how are lipids unique when compared to other macromolecule groups”.
2. Understanding the Four Major Macromolecule Groups
Before diving into the uniqueness of lipids, let’s briefly review the other three major macromolecule groups: carbohydrates, proteins, and nucleic acids.
2.1. Carbohydrates: The Energy Providers and Structural Components
Carbohydrates are primarily known for their role as energy sources. They include sugars, starches, and fibers.
- Structure: Carbohydrates consist of carbon, hydrogen, and oxygen in a 1:2:1 ratio (CH2O)n. They exist as monosaccharides (simple sugars like glucose), disaccharides (two monosaccharides linked together like sucrose), and polysaccharides (long chains of monosaccharides like starch and cellulose).
- Function: Carbohydrates provide quick energy (glucose), store energy (starch and glycogen), and provide structural support in plants (cellulose) and arthropods (chitin).
2.2. Proteins: The Versatile Workhorses
Proteins are the most diverse macromolecules in terms of function. They are involved in virtually every process within a cell.
- Structure: Proteins are polymers of amino acids. Each amino acid contains an amino group (NH2), a carboxyl group (COOH), and a unique R-group that determines its properties. Amino acids are linked together by peptide bonds to form polypeptide chains, which then fold into complex three-dimensional structures.
- Function: Proteins act as enzymes (catalyzing biochemical reactions), hormones (signaling molecules), structural components (collagen, keratin), and antibodies (immune defense).
2.3. Nucleic Acids: The Information Carriers
Nucleic acids, DNA and RNA, store and transmit genetic information.
- Structure: Nucleic acids are polymers of nucleotides. Each nucleotide consists of a pentose sugar (deoxyribose in DNA, ribose in RNA), a phosphate group, and a nitrogenous base (adenine, guanine, cytosine, thymine in DNA; adenine, guanine, cytosine, uracil in RNA). DNA is a double helix, while RNA is typically single-stranded.
- Function: DNA stores genetic information, and RNA is involved in protein synthesis.
3. Lipids: The Hydrophobic Exception
Lipids, also known as fats, are a diverse group of compounds characterized by their hydrophobic nature, meaning they are insoluble in water. This property sets them apart from the other three macromolecule groups, which are generally water-soluble.
3.1. Structure of Lipids
Lipids encompass a wide variety of structures, including triglycerides (fats and oils), phospholipids, steroids, and waxes. While their structures differ, they all share a common feature: a high proportion of nonpolar carbon-carbon and carbon-hydrogen bonds, making them hydrophobic.
- Triglycerides (Fats and Oils): These are composed of a glycerol molecule and three fatty acids. Fatty acids are long hydrocarbon chains with a carboxyl group at one end. They can be saturated (containing only single bonds) or unsaturated (containing one or more double bonds).
- Phospholipids: These are similar to triglycerides, but one fatty acid is replaced by a phosphate group linked to an alcohol. This gives phospholipids a dual nature: the fatty acid tails are hydrophobic, while the phosphate head is hydrophilic.
- Steroids: These have a ring structure, consisting of four fused carbon rings. Examples include cholesterol, testosterone, and estrogen.
- Waxes: These are composed of long-chain fatty acids esterified with long-chain alcohols.
3.2. Functions of Lipids
Lipids perform a variety of essential functions in living organisms:
- Energy Storage: Lipids are an efficient way to store energy due to their high caloric content (9 kcal/gram compared to 4 kcal/gram for carbohydrates and proteins). Triglycerides are the primary form of energy storage in animals.
- Structural Components: Phospholipids are the main components of cell membranes, forming a lipid bilayer that separates the inside of the cell from the outside environment.
- Insulation and Protection: Lipids provide insulation against cold temperatures and protect organs from physical shock.
- Hormone Production: Steroids, such as cholesterol, are precursors to many important hormones that regulate various physiological processes.
- Water Repellency: Waxes provide a protective coating on plant leaves and animal skin, preventing water loss.
4. How Lipids are Unique Compared to Other Macromolecules
Lipids possess several distinguishing characteristics that set them apart from carbohydrates, proteins, and nucleic acids.
4.1. Hydrophobic Nature
The most fundamental difference is their hydrophobic nature. While carbohydrates, proteins, and nucleic acids are generally water-soluble, lipids are not. This is due to their high proportion of nonpolar carbon-carbon and carbon-hydrogen bonds. This insolubility in water has significant implications for their functions, particularly in forming cell membranes and storing energy.
4.2. Structural Diversity
Lipids exhibit a greater structural diversity than carbohydrates. While carbohydrates are primarily based on simple sugar units, lipids encompass a wide range of structures, including linear fatty acids, ring-shaped steroids, and complex phospholipids. This structural diversity allows lipids to perform a wider variety of functions.
4.3. Energy Density
Lipids have a higher energy density than carbohydrates and proteins. They provide more than twice the amount of energy per gram. This makes them an efficient way to store energy for long-term use.
4.4. Lack of a Monomeric Structure
Unlike carbohydrates, proteins, and nucleic acids, lipids do not have a true monomeric structure. While triglycerides are composed of glycerol and fatty acids, these components are not considered monomers in the same way that monosaccharides, amino acids, and nucleotides are. Lipids are more accurately described as being composed of repeating structural units rather than true monomers.
4.5. Different Types of Bonds
The types of bonds present in lipids, especially triglycerides, differ from those in other macromolecules. Ester bonds link fatty acids to glycerol, while glycosidic bonds link monosaccharides in carbohydrates, peptide bonds link amino acids in proteins, and phosphodiester bonds link nucleotides in nucleic acids. These distinct bond types contribute to the unique properties of each macromolecule group.
5. Detailed Comparison: Lipids vs. Carbohydrates, Proteins, and Nucleic Acids
To further highlight the uniqueness of lipids, let’s compare them in detail with the other three macromolecule groups across various criteria.
5.1. Lipids vs. Carbohydrates
| Feature | Lipids | Carbohydrates |
|---|---|---|
| Solubility | Hydrophobic (insoluble in water) | Hydrophilic (soluble in water) |
| Structure | Diverse: triglycerides, phospholipids, steroids, waxes | Primarily based on simple sugar units: monosaccharides, disaccharides, polysaccharides |
| Energy Density | High (9 kcal/gram) | Lower (4 kcal/gram) |
| Monomers | No true monomers | Monosaccharides (glucose, fructose, galactose) |
| Primary Bonds | Ester bonds (triglycerides) | Glycosidic bonds |
| Functions | Energy storage, structural components of membranes, insulation, hormone production | Quick energy, energy storage, structural support in plants and arthropods |
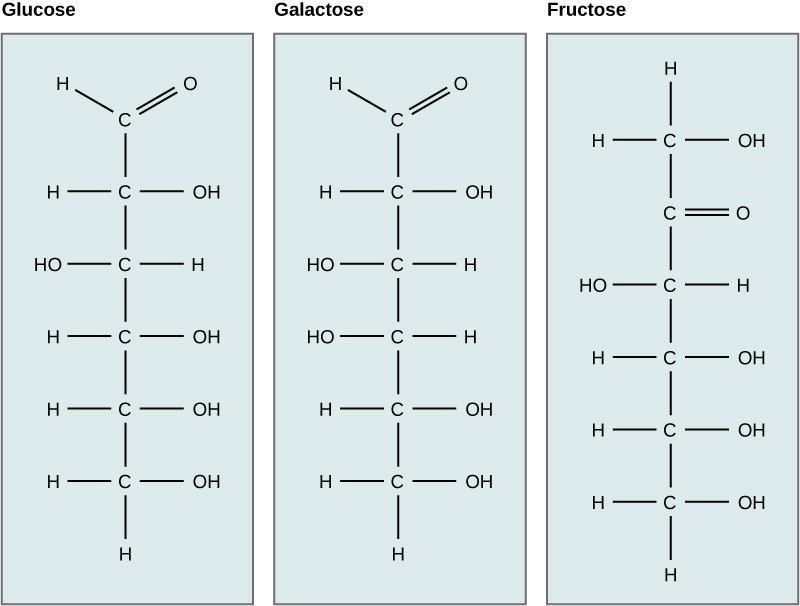

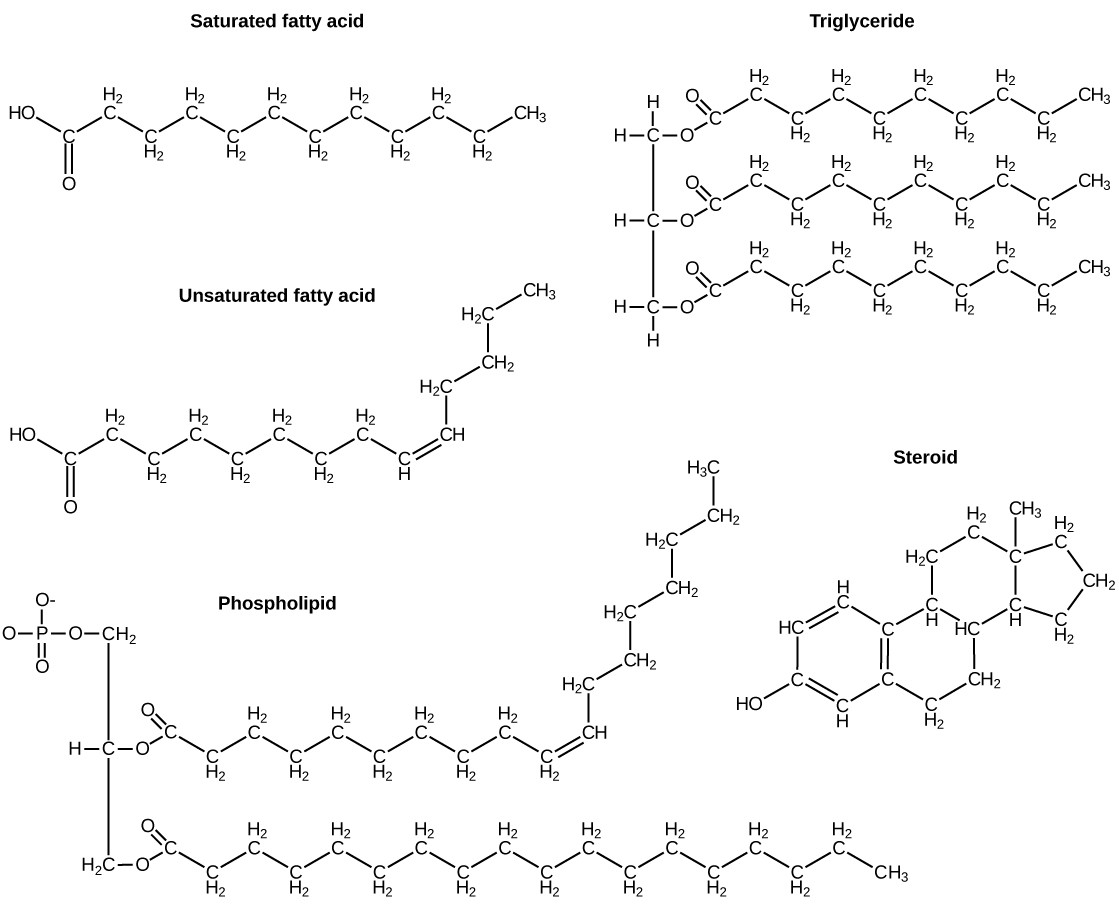
5.2. Lipids vs. Proteins
| Feature | Lipids | Proteins |
|---|---|---|
| Solubility | Hydrophobic (insoluble in water) | Generally hydrophilic (soluble in water), some hydrophobic regions |
| Structure | Diverse: triglycerides, phospholipids, steroids, waxes | Polymers of amino acids, folded into complex three-dimensional structures |
| Energy Density | High (9 kcal/gram) | Moderate (4 kcal/gram) |
| Monomers | No true monomers | Amino acids (20 different types) |
| Primary Bonds | Ester bonds (triglycerides) | Peptide bonds |
| Functions | Energy storage, structural components of membranes, insulation, hormone production | Enzymes, hormones, structural components, antibodies, transport, storage |
5.3. Lipids vs. Nucleic Acids
| Feature | Lipids | Nucleic Acids (DNA and RNA) |
|---|---|---|
| Solubility | Hydrophobic (insoluble in water) | Hydrophilic (soluble in water) |
| Structure | Diverse: triglycerides, phospholipids, steroids, waxes | Polymers of nucleotides, DNA is a double helix, RNA is typically single-stranded |
| Energy Density | High (9 kcal/gram) | Not primarily used for energy storage |
| Monomers | No true monomers | Nucleotides (adenine, guanine, cytosine, thymine/uracil) |
| Primary Bonds | Ester bonds (triglycerides) | Phosphodiester bonds |
| Functions | Energy storage, structural components of membranes, insulation, hormone production | Store genetic information (DNA), protein synthesis (RNA) |
6. The Role of Hydrophobicity in Lipid Function
The hydrophobic nature of lipids is central to their unique functions.
6.1. Formation of Cell Membranes
Phospholipids, with their hydrophobic fatty acid tails and hydrophilic phosphate heads, spontaneously form bilayers in aqueous environments. This lipid bilayer is the foundation of cell membranes, providing a barrier that separates the cell’s interior from the external environment. The hydrophobic core of the bilayer prevents the passage of water-soluble molecules, while allowing the passage of lipid-soluble molecules.
6.2. Efficient Energy Storage
The insolubility of triglycerides in water allows them to be stored in a highly concentrated form without disrupting cellular processes. This makes them an efficient way to store energy for long-term use.
6.3. Insulation and Water Repellency
The hydrophobic nature of lipids provides insulation against cold temperatures by preventing heat loss. It also provides water repellency, protecting organisms from excessive water absorption or loss.
7. The Impact of Lipid Structure on Biological Functions
The specific structure of each type of lipid is closely related to its biological function.
7.1. Triglycerides (Fats and Oils)
The length and saturation of the fatty acid chains in triglycerides affect their physical properties and biological functions. Saturated fats, with their straight chains, pack tightly together and are solid at room temperature. They are primarily used for long-term energy storage. Unsaturated fats, with their bent chains due to double bonds, do not pack tightly and are liquid at room temperature. They are important for cell membrane fluidity and hormone production.
7.2. Phospholipids
The amphipathic nature of phospholipids (having both hydrophobic and hydrophilic regions) is essential for their role in forming cell membranes. The hydrophobic tails create a barrier to water-soluble molecules, while the hydrophilic heads interact with the aqueous environment inside and outside the cell.
7.3. Steroids
The ring structure of steroids provides a rigid framework that allows them to interact with specific receptors and regulate various physiological processes. Cholesterol, for example, is a precursor to many steroid hormones, such as testosterone, estrogen, and cortisol, which regulate reproduction, metabolism, and immune function.
7.4. Waxes
The long-chain fatty acids and alcohols in waxes provide a hydrophobic coating that prevents water loss and protects surfaces from abrasion.
8. The Importance of Lipids in Diet and Health
Lipids play a crucial role in human diet and health.
8.1. Essential Fatty Acids
Some fatty acids, known as essential fatty acids, cannot be synthesized by the human body and must be obtained from the diet. These include omega-3 and omega-6 fatty acids, which are important for brain function, immune function, and cardiovascular health.
8.2. Cholesterol and Heart Health
Cholesterol is an essential component of cell membranes and a precursor to many hormones. However, high levels of cholesterol in the blood can lead to the formation of plaque in arteries, increasing the risk of heart disease.
8.3. Saturated vs. Unsaturated Fats
Saturated fats, found primarily in animal products, can raise blood cholesterol levels and increase the risk of heart disease. Unsaturated fats, found primarily in plant oils and fish, can help lower blood cholesterol levels and reduce the risk of heart disease.
8.4. Trans Fats
Trans fats are unsaturated fats that have been artificially hydrogenated to make them solid at room temperature. They have been shown to raise LDL (“bad”) cholesterol levels and lower HDL (“good”) cholesterol levels, increasing the risk of heart disease.
9. Common Misconceptions About Lipids
There are several common misconceptions about lipids that should be addressed.
9.1. All Fats are Bad
Not all fats are bad. In fact, some fats, such as unsaturated fats and essential fatty acids, are essential for health. The key is to consume fats in moderation and to choose healthy fats over unhealthy fats.
9.2. Lipids Only Serve as Energy Storage
While energy storage is a major function of lipids, they also play many other important roles, including forming cell membranes, providing insulation, and producing hormones.
9.3. Cholesterol is Always Harmful
Cholesterol is essential for cell membrane structure and hormone production. It is only harmful when levels in the blood become too high, leading to plaque formation in arteries.
10. The Role of Lipids in Various Organisms
Lipids play diverse roles across different organisms.
10.1. Plants
In plants, lipids are crucial for energy storage in seeds, providing the energy needed for germination and early growth. Waxes on leaf surfaces prevent water loss, and phospholipids form the membranes of chloroplasts and other organelles.
10.2. Animals
In animals, lipids provide long-term energy storage in adipose tissue, insulate against cold temperatures, and protect organs from physical shock. Phospholipids form cell membranes, and steroids regulate various physiological processes.
10.3. Microorganisms
In microorganisms, lipids form cell membranes and provide energy storage. Some bacteria have unique lipids in their cell walls that protect them from harsh environments.
11. Technological and Industrial Applications of Lipids
Lipids have numerous technological and industrial applications.
11.1. Food Industry
Lipids are used in the food industry for various purposes, including:
- Cooking Oils: Triglycerides are used as cooking oils for frying, baking, and sautéing.
- Emulsifiers: Phospholipids are used as emulsifiers to stabilize mixtures of oil and water, such as in mayonnaise and salad dressings.
- Flavor Enhancers: Lipids can enhance the flavor and texture of foods.
11.2. Cosmetics and Pharmaceuticals
Lipids are used in cosmetics and pharmaceuticals for various purposes, including:
- Emollients: Lipids are used as emollients to soften and moisturize the skin.
- Drug Delivery: Liposomes, which are made of phospholipids, are used to deliver drugs to specific cells or tissues.
- Hormone Production: Steroids are used in hormone replacement therapy and as anti-inflammatory drugs.
11.3. Biofuels
Lipids can be converted into biofuels, such as biodiesel, which can be used as an alternative to petroleum-based fuels.
12. Emerging Research in Lipid Biology
Lipid biology is a rapidly advancing field with many exciting areas of research.
12.1. Lipidomics
Lipidomics is the study of all lipids in a biological system. It aims to identify and quantify all the different types of lipids present in a cell, tissue, or organism. This information can be used to understand the role of lipids in various biological processes and diseases.
12.2. Lipid Rafts
Lipid rafts are specialized microdomains in cell membranes that are enriched in certain lipids, such as cholesterol and sphingolipids. They are thought to play a role in signal transduction, membrane trafficking, and protein sorting.
12.3. Non-Alcoholic Fatty Liver Disease (NAFLD)
NAFLD is a common liver disease characterized by the accumulation of fat in the liver. It is often associated with obesity, diabetes, and other metabolic disorders. Research is ongoing to understand the causes and mechanisms of NAFLD and to develop effective treatments.
13. Conclusion: Embracing the Uniqueness of Lipids
In conclusion, lipids are unique macromolecules with distinct properties and functions that set them apart from carbohydrates, proteins, and nucleic acids. Their hydrophobic nature, structural diversity, high energy density, and lack of a true monomeric structure contribute to their essential roles in energy storage, cell membrane structure, insulation, hormone production, and more. Understanding the uniqueness of lipids is crucial for comprehending their importance in biology, health, and technology.
For more in-depth comparisons and information on lipids and other macromolecules, visit COMPARE.EDU.VN. Our comprehensive resources can help you make informed decisions about nutrition, health, and related topics.
14. COMPARE.EDU.VN: Your Partner in Informed Decisions
Navigating the complexities of biological molecules and their impact on health can be challenging. COMPARE.EDU.VN simplifies this process by providing detailed, objective comparisons. Whether you’re a student, a healthcare professional, or simply someone interested in making informed decisions about your health, our website offers the resources you need.
14.1. Why Choose COMPARE.EDU.VN?
- Comprehensive Comparisons: We offer side-by-side comparisons of various topics, including the differences between lipids, carbohydrates, proteins, and nucleic acids.
- Objective Information: Our content is based on scientific research and expert analysis, ensuring you receive unbiased information.
- User-Friendly Interface: Our website is designed to be easy to navigate, allowing you to quickly find the information you need.
- Regular Updates: We continuously update our content to reflect the latest research and developments in the field.
14.2. Explore Our Resources
Visit COMPARE.EDU.VN to explore our extensive library of comparisons and articles. Learn about:
- The different types of lipids and their health effects
- The role of lipids in various diseases
- The latest research in lipid biology
- Practical tips for incorporating healthy fats into your diet
15. Call to Action
Ready to make informed decisions about your health and nutrition? Visit COMPARE.EDU.VN today to explore our comprehensive comparisons and resources.
Address: 333 Comparison Plaza, Choice City, CA 90210, United States
WhatsApp: +1 (626) 555-9090
Website: COMPARE.EDU.VN
Unlock the power of knowledge with COMPARE.EDU.VN and take control of your health journey now
16. Frequently Asked Questions (FAQ) about Lipids
16.1. What are lipids?
Lipids are a diverse group of organic compounds that are insoluble in water. They include fats, oils, waxes, phospholipids, and steroids.
16.2. What are the main functions of lipids?
Lipids serve several important functions, including energy storage, forming cell membranes, providing insulation, and producing hormones.
16.3. How are lipids different from carbohydrates, proteins, and nucleic acids?
Lipids are hydrophobic (insoluble in water), have a higher energy density, and do not have a true monomeric structure, unlike carbohydrates, proteins, and nucleic acids.
16.4. What are triglycerides?
Triglycerides are the main type of fat found in the body. They are composed of a glycerol molecule and three fatty acids.
16.5. What are saturated and unsaturated fats?
Saturated fats contain only single bonds in their fatty acid chains, while unsaturated fats contain one or more double bonds. Saturated fats are typically solid at room temperature, while unsaturated fats are liquid.
16.6. What are phospholipids?
Phospholipids are lipids that contain a phosphate group. They are the main components of cell membranes.
16.7. What are steroids?
Steroids are lipids with a ring structure. They include cholesterol and various hormones, such as testosterone and estrogen.
16.8. What are essential fatty acids?
Essential fatty acids are fatty acids that cannot be synthesized by the human body and must be obtained from the diet. They include omega-3 and omega-6 fatty acids.
16.9. How do lipids contribute to heart health?
Consuming healthy fats, such as unsaturated fats, can help lower blood cholesterol levels and reduce the risk of heart disease. Limiting saturated and trans fats is important for maintaining heart health.
16.10. Where can I find more information about lipids and nutrition?
Visit compare.edu.vn for comprehensive comparisons, resources, and the latest research on lipids and their role in health and nutrition. Our expert-backed information is designed to help you make informed decisions and optimize your well-being.
17. References
- Nelson, D. L., & Cox, M. M. (2017). Lehninger principles of biochemistry (7th ed.). W. H. Freeman.
- Berg, J. M., Tymoczko, J. L., & Stryer, L. (2012). Biochemistry (7th ed.). W. H. Freeman.
- Lodish, H., Berk, A., Zipursky, S. L., et al. (2000). Molecular cell biology (4th ed.). W. H. Freeman.
- Wilkerson, Suzanne, and Charles Molnar. “Through the Indigenous Lens.” Biology, Open Textbook Library, 2021, opentextbc.ca/biology/. Accessed 25, May 2024.
