X-rays and ultraviolet (UV) rays are both forms of electromagnetic radiation, populating different segments of the electromagnetic spectrum. While both are invisible to the human eye and possess energy that can interact with matter, they exhibit key differences in their properties, origins, applications, and potential hazards. Understanding these distinctions is crucial in various fields, from medicine and astronomy to environmental science and daily life.
Unveiling X-Rays: High Energy and Penetration
X-rays are characterized by their high energy and short wavelengths, ranging from 0.03 to 3 nanometers. Often described in terms of their energy due to these minuscule wavelengths, some x-rays are even smaller than a single atom. This high energy endows x-rays with significant penetrating power, allowing them to pass through soft tissues but be absorbed by denser materials like bones and metals.
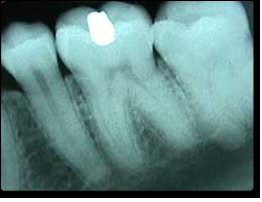
The discovery of X-rays is attributed to Wilhelm Conrad Roentgen in 1895. He famously demonstrated their ability to visualize bones within the human body, revolutionizing medical diagnostics. In medical applications, X-rays are directed through the body, and the differential absorption by tissues creates shadow images on X-ray sensitive film or digital detectors, revealing fractures, foreign objects, and other internal conditions.
Beyond medicine, X-rays are vital in astronomy for studying extremely hot and energetic celestial objects. The Sun’s corona, for instance, emits primarily X-rays due to its high temperature. Satellites equipped with X-ray detectors, like Japan’s Hinode spacecraft, capture these emissions, providing insights into solar flares, sunspot activity, and energy flows within the corona.
Objects emitting X-rays are typically at temperatures of millions of degrees Celsius, such as pulsars, supernova remnants, and the accretion disks around black holes. X-ray telescopes in space collect these photons, and by analyzing their energy, time, and direction, scientists can deduce the composition, temperature, and density of distant cosmic environments. Space-based telescopes are necessary as Earth’s atmosphere blocks X-ray radiation.
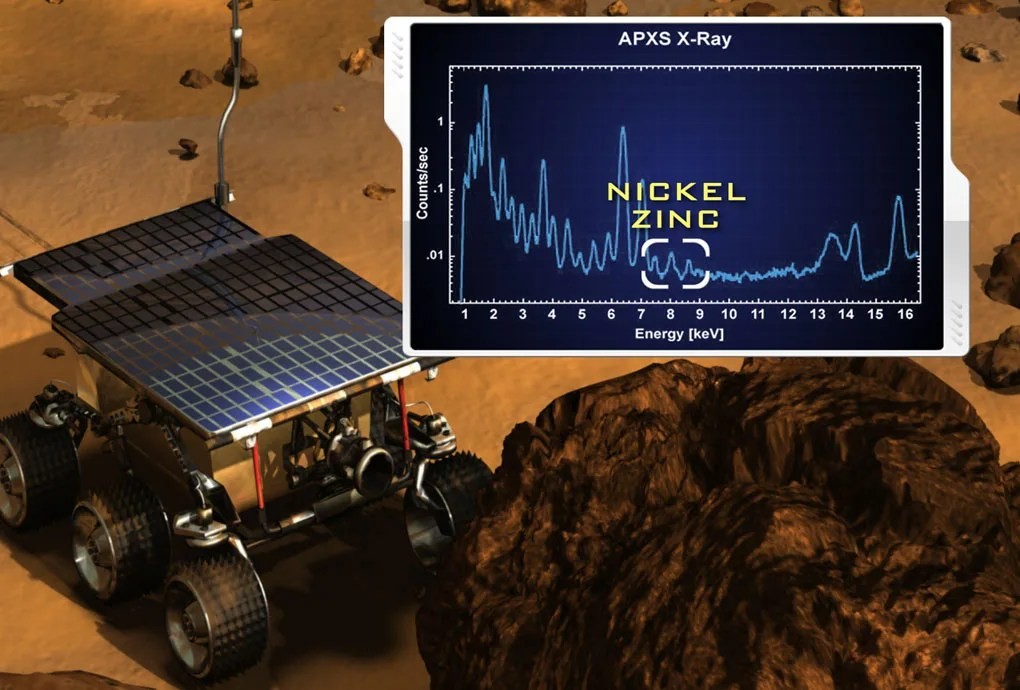
Even planetary science benefits from X-rays. NASA’s Mars Exploration Rover Spirit used an Alpha Proton X-Ray Spectrometer (APXS) to identify elements like zinc and nickel in Martian rocks, aiding in understanding the planet’s geological composition.
Understanding UV Rays: Lower Energy but Still Potent
Ultraviolet (UV) rays, while still part of the electromagnetic spectrum, possess lower energy and longer wavelengths compared to X-rays, ranging from approximately 10 to 400 nanometers. UV radiation is further categorized into UVA, UVB, and UVC based on wavelength, each with varying energy levels and impacts.
UVA rays have the longest wavelengths and are least energetic, penetrating deeply into the skin and contributing to tanning and skin aging. UVB rays have shorter wavelengths and higher energy, responsible for sunburn and a major factor in skin cancer. UVC rays are the most energetic but are mostly absorbed by the Earth’s atmosphere and do not pose a significant threat at ground level.
The primary source of UV radiation is the Sun. While essential for Vitamin D synthesis in humans and various biological processes, excessive exposure to UV radiation can be harmful. UV radiation’s energy is sufficient to damage DNA and living cells, leading to sunburn, premature aging, cataracts, and skin cancer.
Despite the risks, UV rays have beneficial applications. UV sterilization is widely used to disinfect water, air, and surfaces due to UV radiation’s ability to kill microorganisms. In medicine, controlled UV exposure is used to treat certain skin conditions like psoriasis and vitiligo. Furthermore, UV lamps are used in various industrial processes, such as curing adhesives and inks.
X-Rays vs. UV Rays: Key Differences and Similarities
| Feature | X-Rays | UV Rays |
|---|---|---|
| Energy Level | High | Lower |
| Wavelength | Very Short (0.03 – 3 nanometers) | Longer (10 – 400 nanometers) |
| Penetration | High, penetrates soft tissue, blocked by dense materials | Lower, absorbed by skin and some materials |
| Primary Sources | Extremely hot objects (space), X-ray tubes | Sun, UV lamps |
| Applications | Medical imaging, astronomy, material analysis, security scanning | Sterilization, Vitamin D synthesis, dermatology treatments, industrial curing |
| Hazards | Ionizing radiation, cancer risk with high exposure | Sunburn, skin cancer, cataracts, premature aging |


Similarities:
- Both are forms of electromagnetic radiation, part of the broader electromagnetic spectrum.
- Both are invisible to the human eye.
- Both can interact with matter and have applications in science, technology, and medicine.
- Both can be harmful to living organisms in excessive doses.
Conclusion: Navigating the Electromagnetic Spectrum
X-rays and UV rays, while both electromagnetic radiation, occupy distinct positions within the spectrum, leading to significant differences in their energy, properties, and interactions with matter. X-rays, with their high energy and penetrating power, are invaluable for peering inside the human body and exploring the energetic cosmos. UV rays, though less energetic, play crucial roles in both biological processes and technological applications, while also posing risks with overexposure. Understanding the nuances between these radiation types is essential for harnessing their benefits safely and mitigating potential hazards in our daily lives and across scientific disciplines.