Understanding the role of solvents in UV Vis spectroscopy is crucial for accurate measurements. Different solvents can significantly impact the absorption spectrum of a compound. This article explores the key factors to consider when comparing two different UV Vis solvents and provides examples to illustrate these concepts.
Solvent Polarity and Its Effects on UV Vis Spectra
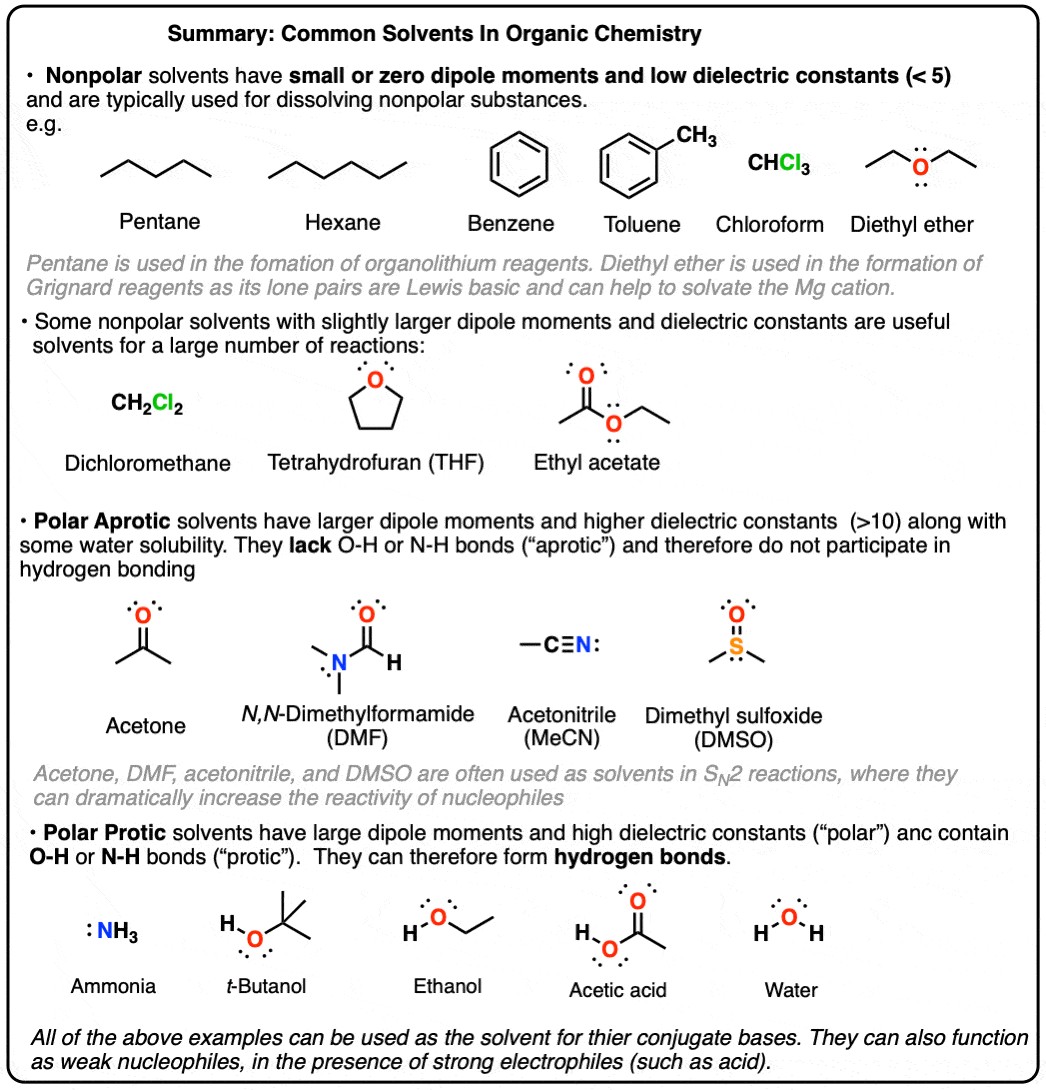
Solvent polarity plays a critical role in UV Vis spectroscopy. Polar solvents can interact with the analyte molecules through dipole-dipole interactions or hydrogen bonding, leading to changes in the energy levels of the electronic transitions. This can result in shifts in the absorption maxima (λmax) and changes in the intensity of the absorption bands. Generally:
- Polar protic solvents: These solvents (e.g., water, methanol, ethanol) can form strong hydrogen bonds with analyte molecules, often leading to significant spectral shifts, particularly for compounds with polar functional groups capable of hydrogen bonding.
- Polar aprotic solvents: These solvents (e.g., acetonitrile, DMSO, DMF) exhibit dipole-dipole interactions but lack O-H or N-H bonds for hydrogen bonding. Their impact on UV Vis spectra is generally less pronounced than that of polar protic solvents.
- Nonpolar solvents: These solvents (e.g., hexane, cyclohexane) have minimal interaction with analyte molecules. They are preferred when minimal solvent interference is desired. However, they may not dissolve all analytes.
Solvent Cut-off Wavelength
Each solvent has a characteristic cut-off wavelength below which it absorbs strongly in the UV region. Choosing a solvent with a cut-off wavelength lower than the expected absorption range of the analyte is essential to avoid interference. For instance:
- Water: has a cut-off wavelength around 190 nm.
- Acetonitrile: has a cut-off wavelength around 190 nm.
- Methanol: has a cut-off wavelength around 205 nm.
- Hexane: has a cut-off wavelength around 210 nm.
Comparing Two Solvents: A Practical Example
Let’s compare two common solvents: methanol (polar protic) and acetonitrile (polar aprotic).
If analyzing a ketone, methanol’s ability to hydrogen bond with the carbonyl oxygen might cause a bathochromic shift (redshift) in the n→π* transition. In contrast, acetonitrile, lacking hydrogen bonding capability, would likely induce a smaller shift or even a hypsochromic shift (blueshift) due to its polar nature stabilizing the ground state more than the excited state.
Solvent Effects on Specific Chromophores
Different chromophores exhibit varying sensitivities to solvent polarity. For example, conjugated systems often show more significant solvatochromism than isolated double bonds.
Conclusion: Selecting the Appropriate Solvent
When comparing UV Vis solvents, consider polarity, cut-off wavelength, and potential specific interactions with the analyte. Choosing the right solvent is crucial for obtaining accurate and meaningful spectral data. Careful consideration of these factors ensures reliable results in UV Vis spectroscopy experiments. Always consult relevant literature and spectral databases for guidance on solvent selection for specific analytes.