Are Hydrogen Bonds Weak Compared To Covalent bonds? This question is often asked in introductory chemistry, and while the general answer is yes, COMPARE.EDU.VN explores the nuances and complexities that blur the lines between these two fundamental types of chemical bonds. This article provides a comprehensive comparison of hydrogen bonds and covalent bonds, delving into their definitions, strengths, formation, and roles in various chemical and biological systems. We’ll also examine recent research that challenges traditional views on hydrogen bond strength, touching upon intermolecular forces and electrostatic interactions.
1. Understanding Covalent Bonds
Covalent bonds are a cornerstone of chemistry, representing the sharing of electrons between atoms to achieve stability. These bonds are responsible for the structure of countless molecules, from simple diatomic gases to complex organic compounds.
1.1. Definition of Covalent Bonds
A covalent bond occurs when atoms share one or more pairs of electrons to achieve a stable electron configuration, typically resembling that of a noble gas. This sharing of electrons leads to a mutual attraction between the positively charged nuclei and the negatively charged shared electrons, effectively holding the atoms together.
1.2. Formation of Covalent Bonds
Covalent bonds typically form between two nonmetal atoms. The process involves the overlap of atomic orbitals, creating a region of high electron density between the nuclei. This shared electron density reduces the overall energy of the system, leading to a stable bond.
1.3. Strength of Covalent Bonds
Covalent bonds are generally strong, with bond energies ranging from 150 to 1100 kJ/mol. The strength of a covalent bond depends on factors such as the number of shared electrons (single, double, or triple bonds), the electronegativity difference between the bonded atoms, and the size of the atoms involved.
1.4. Types of Covalent Bonds
There are two main types of covalent bonds: polar and nonpolar.
- Nonpolar Covalent Bonds: These bonds form when electrons are shared equally between two atoms with similar electronegativity values. Examples include bonds between two identical atoms, such as in H₂, O₂, and Cl₂.
- Polar Covalent Bonds: These bonds form when electrons are shared unequally between two atoms with different electronegativity values. The more electronegative atom attracts the electron density, resulting in a partial negative charge (δ-) on that atom and a partial positive charge (δ+) on the other atom. Examples include bonds in H₂O, HF, and NH₃.
2. Exploring Hydrogen Bonds
Hydrogen bonds are a unique type of intermolecular force that plays a critical role in various chemical and biological processes. They are weaker than covalent bonds but are essential for stabilizing the structures of water, proteins, DNA, and other important molecules.
2.1. Definition of Hydrogen Bonds
A hydrogen bond is an electrostatic attraction between a hydrogen atom covalently bonded to a highly electronegative atom (such as oxygen, nitrogen, or fluorine) and another electronegative atom in the same or a different molecule. The hydrogen atom carries a partial positive charge (δ+), while the electronegative atom carries a partial negative charge (δ-), leading to an attractive force between them.
2.2. Formation of Hydrogen Bonds
Hydrogen bonds form when a hydrogen atom is bonded to a highly electronegative atom, creating a significant dipole moment. This dipole moment allows the hydrogen atom to be attracted to another electronegative atom with a lone pair of electrons. The strength of a hydrogen bond depends on the distance and angle between the interacting atoms.
2.3. Strength of Hydrogen Bonds
Hydrogen bonds are weaker than covalent bonds, with bond energies typically ranging from 4 to 50 kJ/mol. However, the cumulative effect of many hydrogen bonds can be significant, contributing to the overall stability of a structure.
2.4. Types of Hydrogen Bonds
Hydrogen bonds can be classified as either intermolecular or intramolecular.
- Intermolecular Hydrogen Bonds: These bonds form between different molecules, such as between water molecules in liquid water or ice.
- Intramolecular Hydrogen Bonds: These bonds form within the same molecule, such as in proteins and DNA, helping to stabilize their three-dimensional structures.
3. Key Differences Between Hydrogen Bonds and Covalent Bonds
While both hydrogen bonds and covalent bonds involve attractive forces between atoms, they differ significantly in their nature, strength, and formation. The table below summarizes the key differences.
| Feature | Covalent Bond | Hydrogen Bond |
|---|---|---|
| Nature | Sharing of electrons | Electrostatic attraction |
| Strength | Strong (150-1100 kJ/mol) | Weak (4-50 kJ/mol) |
| Formation | Between two nonmetal atoms | Between H bonded to electronegative atom and another electronegative atom |
| Bonding Atoms | Nonmetal-nonmetal | H-O, H-N, or H-F…O, N, or F |
| Location | Within molecules | Between molecules (intermolecular) or within a molecule (intramolecular) |
4. Are Hydrogen Bonds Really That Weak? Exploring the Continuum
The textbook picture of hydrogen bonds as simply weak electrostatic interactions is being challenged by recent research. Scientists are discovering that under certain conditions, hydrogen bonds can exhibit characteristics more akin to covalent bonds, blurring the lines between these two types of interactions.
4.1. The F-H-F- Ion: A Case Study
One compelling example of this blurring is the F-H-F- ion. In this ion, a fluorine anion (F-) forms a hydrogen bond with the hydrogen atom in a covalent HF molecule. Spectroscopic experiments and computer simulations have revealed that the hydrogen bond in F-H-F- can be remarkably strong, approaching the strength of a covalent bond.
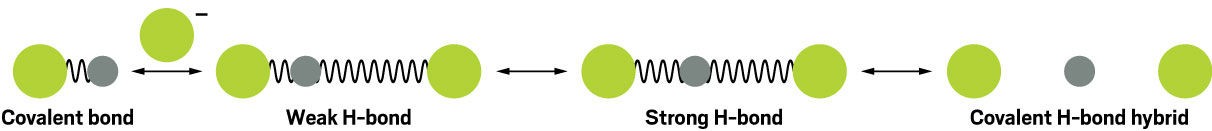 Diagram of transition from covalent HF to covalent-like strong hydrogen bonding in F-H-F anion, illustrating the shift in vibrational behavior from hydrogen to covalent bonds
Diagram of transition from covalent HF to covalent-like strong hydrogen bonding in F-H-F anion, illustrating the shift in vibrational behavior from hydrogen to covalent bonds
4.2. Factors Influencing Hydrogen Bond Strength
Several factors can influence the strength of hydrogen bonds, including:
- Distance: Shorter hydrogen bonds are generally stronger due to increased electrostatic attraction.
- Angle: Linear hydrogen bonds (where the hydrogen atom is aligned directly between the two electronegative atoms) are stronger than bent hydrogen bonds.
- Environment: The surrounding environment, such as the presence of water molecules or other ions, can affect the strength of hydrogen bonds.
4.3. The Continuum of Bonding
The research on the F-H-F- ion suggests that there is not a clear-cut distinction between hydrogen bonds and covalent bonds, but rather a continuum of bonding interactions. At one end of the spectrum are weak, purely electrostatic hydrogen bonds, while at the other end are strong, covalent-like hydrogen bonds with significant electron sharing.
4.4. Implications for Chemistry and Biology
This new understanding of hydrogen bonding has significant implications for various fields of chemistry and biology. For example, it may help scientists better understand the role of hydrogen bonds in enzyme catalysis, protein folding, and DNA replication.
5. The Role of Hydrogen Bonds in Biological Systems
Hydrogen bonds are essential for life as we know it, playing a critical role in the structure and function of biological molecules.
5.1. Water: The Solvent of Life
Water’s unique properties, such as its high boiling point and surface tension, are due to the extensive network of hydrogen bonds between water molecules. These hydrogen bonds are also responsible for water’s ability to act as a solvent for polar and ionic compounds, making it an ideal medium for biological reactions.
5.2. DNA: The Blueprint of Life
Hydrogen bonds play a crucial role in holding together the two strands of the DNA double helix. Specifically, hydrogen bonds form between complementary base pairs (adenine with thymine and guanine with cytosine), ensuring the accurate replication and transmission of genetic information.
5.3. Proteins: The Workhorses of the Cell
Proteins are complex molecules that perform a wide variety of functions in the cell, from catalyzing biochemical reactions to transporting molecules across cell membranes. Hydrogen bonds are essential for stabilizing the three-dimensional structures of proteins, which are critical for their function.
6. Comparing the Impact: Hydrogen vs. Covalent Bonds
To truly appreciate the distinctions and shared importance of hydrogen and covalent bonds, let’s delve into a side-by-side comparison of their impact across various chemical phenomena.
6.1. Boiling Point
Covalent bonds determine the boiling point of a substance. Substances with strong covalent networks, such as diamond or silicon dioxide, have extremely high boiling points, while those held by weaker intermolecular forces have lower boiling points. Hydrogen bonds play a supporting role, increasing boiling points.
6.2. Solubility
Covalent bonds, specifically their polarity, influence solubility, because “like dissolves like.” Hydrogen bonds enhance water solubility for molecules containing hydrogen bond donors and acceptors, such as alcohols and sugars.
6.3. Molecular Shape
Covalent bonds define the fundamental shape of the molecule, dictating bond lengths and angles. Hydrogen bonds fine-tune the shape, especially in larger molecules like proteins, where they stabilize secondary structures such as alpha-helices and beta-sheets.
6.4. Chemical Reactions
Covalent bonds must be broken and formed during chemical reactions, requiring significant energy input. Hydrogen bonds can be broken and reformed much more easily, enabling dynamic processes such as enzyme-substrate interactions and protein folding.
6.5. Structural Stability
Covalent bonds ensure the basic structural integrity of molecules, while hydrogen bonds are critical for maintaining the higher-order structures of macromolecules such as DNA and proteins, as well as ice and water.
7. Modern Research on Hydrogen Bonds
Modern research techniques and theoretical calculations are continually enhancing our understanding of hydrogen bonds. Let’s look at some groundbreaking recent findings.
7.1. Spectroscopic Methods
Advanced spectroscopic methods, such as femtosecond infrared spectroscopy, provide direct insight into the dynamics of hydrogen bonds, including bond lifetimes, vibrational frequencies, and structural fluctuations.
7.2. Computational Modeling
Sophisticated computational modeling techniques, such as molecular dynamics simulations, allow researchers to simulate the behavior of hydrogen-bonded systems at the atomic level, providing a detailed understanding of their properties.
7.3. Experimental Findings
Experimental studies are revealing new examples of strong hydrogen bonds and challenging the traditional view of hydrogen bonds as weak interactions.
8. Optimizing Outcomes: Leveraging Bond Attributes for Design
In engineering new substances and systems, chemists and materials scientists utilize knowledge of the characteristics of hydrogen and covalent bonds.
8.1. Polymer Science
Polymers gain mechanical strength and thermal stability by combining strong covalent backbones with intermolecular hydrogen bonding, enhancing impact resistance and flexibility.
8.2. Drug Design
Pharmaceutical scientists use hydrogen bonding in molecular docking to enhance the specificity of drug molecules for their biological targets.
8.3. Nanomaterials
Nanomaterials like nanotubes and nanowires self-assemble due to hydrogen bonds, thus opening up new possibilities for electronic and sensing applications.
9. Case Studies: Hydrogen and Covalent Bonds in Action
To provide real-world contexts, let’s explore case studies where hydrogen and covalent bonds are at play in phenomena we encounter daily.
9.1. Water and Ice
Water’s characteristics are heavily affected by the interactions of hydrogen bonds that result in ice. Ice exhibits a structured network of hydrogen bonds that leads to its decreased density compared to liquid water, making it possible for aquatic life to exist during cold conditions.
9.2. Protein Folding
The correct folding of proteins depends on complicated interactions between hydrogen bonds, covalent bonds (such as disulfide bridges), and hydrophobic interactions. Misfolded proteins can cause a variety of illnesses.
9.3. Enzyme Catalysis
Enzymes reduce activation energies by precisely orienting substrates using hydrogen bonds and other non-covalent interactions, considerably speeding up reaction rates.
10. FAQ About Hydrogen Bonds and Covalent Bonds
Here are some frequently asked questions about hydrogen bonds and covalent bonds:
- What is the primary difference between a hydrogen bond and a covalent bond? A covalent bond involves sharing electrons between atoms, while a hydrogen bond is an electrostatic attraction between a hydrogen atom and an electronegative atom.
- Are hydrogen bonds always weaker than covalent bonds? Yes, hydrogen bonds are generally weaker than covalent bonds, but under certain conditions, they can be surprisingly strong.
- What are the key factors that influence the strength of hydrogen bonds? Distance, angle, and the surrounding environment all influence the strength of hydrogen bonds.
- Why are hydrogen bonds important in biological systems? Hydrogen bonds are essential for the structure and function of water, DNA, proteins, and other important biological molecules.
- Can hydrogen bonds form between any two molecules? No, hydrogen bonds require a hydrogen atom bonded to a highly electronegative atom (O, N, or F) and another electronegative atom with a lone pair of electrons.
- What is the difference between intermolecular and intramolecular hydrogen bonds? Intermolecular hydrogen bonds form between different molecules, while intramolecular hydrogen bonds form within the same molecule.
- How do hydrogen bonds contribute to the properties of water? Hydrogen bonds are responsible for water’s high boiling point, surface tension, and ability to act as a solvent.
- What role do hydrogen bonds play in DNA structure? Hydrogen bonds hold together the two strands of the DNA double helix, ensuring accurate replication and transmission of genetic information.
- How do hydrogen bonds contribute to protein folding? Hydrogen bonds stabilize the three-dimensional structures of proteins, which are critical for their function.
- Are hydrogen bonds considered intermolecular forces? Yes, hydrogen bonds are a type of intermolecular force, specifically a dipole-dipole interaction.
11. Conclusion: Appreciating the Interplay of Chemical Bonds
While covalent bonds provide the fundamental framework of molecules, hydrogen bonds fine-tune their properties and interactions. Understanding the differences and similarities between these two types of bonds is crucial for comprehending the behavior of matter at the molecular level.
Whether you’re comparing different molecular structures, exploring the properties of materials, or delving into the intricacies of biological systems, the information provided by COMPARE.EDU.VN empowers you to make informed decisions. Our platform provides comprehensive comparisons and analyses, helping you to navigate the complexities of chemistry and other fields.
Ready to explore further? Visit COMPARE.EDU.VN today to discover more detailed comparisons and insights that can help you make informed choices.
Contact us:
Address: 333 Comparison Plaza, Choice City, CA 90210, United States
Whatsapp: +1 (626) 555-9090
Website: compare.edu.vn