A Comparative Review Of Neutrophil Extracellular Traps In Sepsis reveals their dual role in both protecting against and exacerbating the condition. At COMPARE.EDU.VN, we synthesize research to provide a clear understanding of these complex immune structures and their impact on sepsis pathology, offering insights into potential therapeutic interventions and a comprehensive analysis. NETs function, formation mechanism and clinical significance.
1. Introduction to Sepsis and Neutrophil Extracellular Traps (NETs)
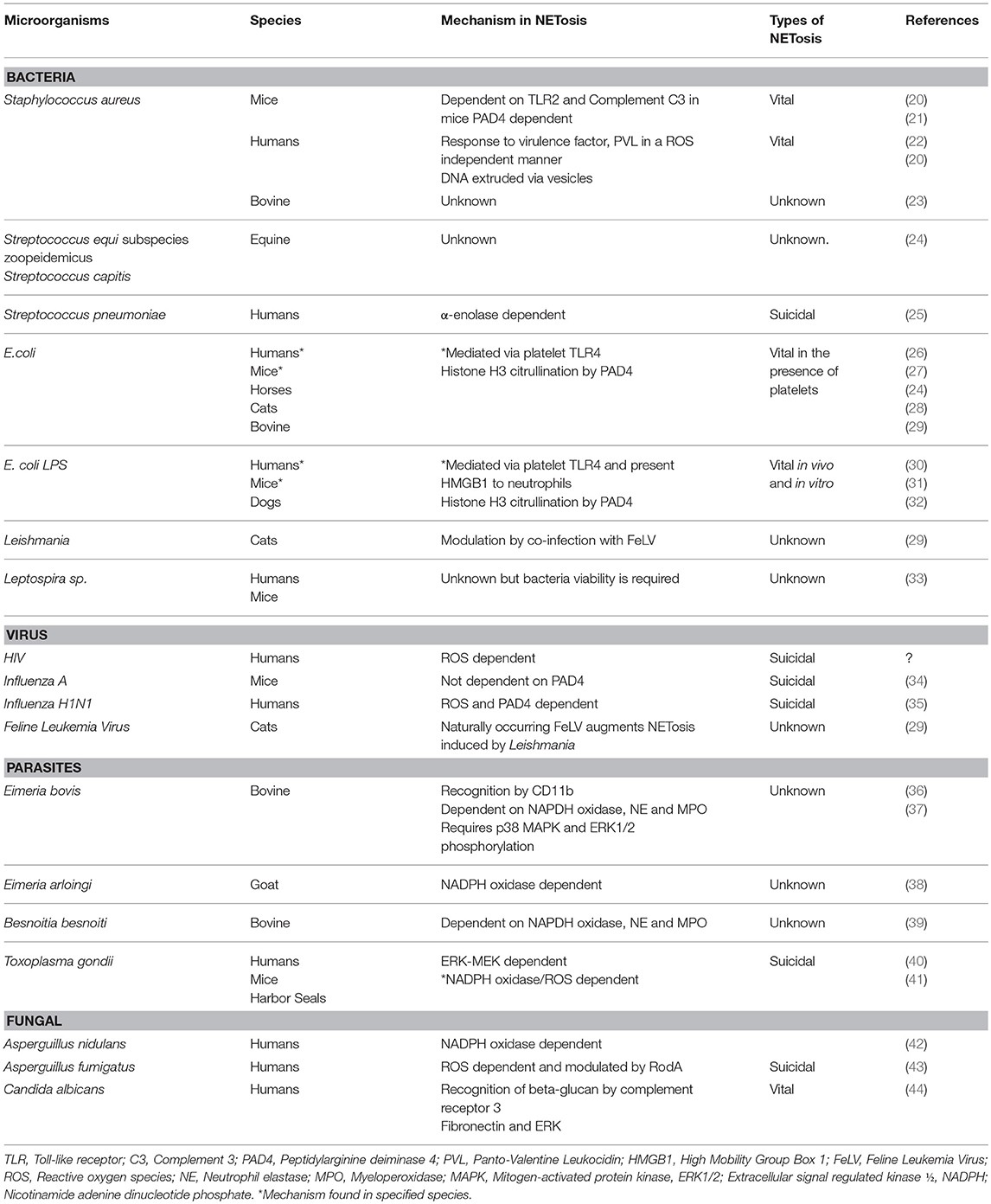
Sepsis remains a leading cause of morbidity and mortality in both human and veterinary medicine, despite advances in treatment protocols. Characterized by a dysregulated host response to infection, sepsis leads to life-threatening organ dysfunction, often culminating in multiple organ failure. Neutrophils, the primary effector cells of the innate immune system, play a crucial role in combating invading pathogens through phagocytosis, degranulation, and the production of reactive oxygen species (ROS). However, in response to infection and inflammation, neutrophils can also release neutrophil extracellular traps (NETs), composed of DNA, histones, and antimicrobial proteins, which have been identified across various species. These NETs, while initially protective by trapping and neutralizing pathogens, can paradoxically contribute to the pathogenesis of sepsis through mechanisms such as promoting thrombosis and inducing organ damage.
1.1. Defining Sepsis and Its Impact
Sepsis, recognized as a critical healthcare challenge, is defined by life-threatening organ dysfunction resulting from a dysregulated host response to infection. This condition affects millions globally, leading to significant morbidity and mortality in both human and animal populations. The transition from localized infection to systemic inflammation and organ failure underscores the complexity of sepsis, making early diagnosis and intervention critical.
1.2. The Role of Neutrophils in Immune Defense
Neutrophils are essential components of the innate immune system, quickly mobilized to sites of infection where they eliminate pathogens through several mechanisms. These include phagocytosis, where pathogens are engulfed and destroyed; degranulation, the release of antimicrobial substances; and the production of ROS to kill pathogens directly. These functions are crucial for initial defense, yet the release of NETs introduces a complex dynamic to the immune response in sepsis.
1.3. What are Neutrophil Extracellular Traps (NETs)?
Neutrophil extracellular traps (NETs) are web-like structures released by neutrophils, composed of DNA, histones, and antimicrobial proteins. These structures are designed to trap and kill pathogens extracellularly. The discovery of NETs has expanded our understanding of neutrophil function, revealing their active role in modulating the immune response.
1.4. The Dual Role of NETs in Sepsis
NETs serve a dual role in sepsis: initially, they are protective by trapping and neutralizing pathogens, thus preventing their spread. However, excessive or dysregulated NET formation can contribute to the pathogenesis of sepsis. This includes promoting thrombosis, exacerbating inflammation, and causing direct damage to organs.
2. The Formation of Neutrophil Extracellular Traps (NETosis)
NETosis, the process by which neutrophils release NETs, is a complex and regulated form of cell death distinct from apoptosis or necrosis. It involves a series of intracellular events leading to the decondensation of chromatin, mixing of nuclear and cytoplasmic contents, and eventual release of DNA into the extracellular space. Understanding NETosis and what triggers is essential for modulating NET-related effects in sepsis.
2.1. Defining NETosis: The Process of NET Formation
NETosis is a unique cell death program that neutrophils undergo to produce NETs. This process involves the activation of neutrophils by various stimuli, leading to changes within the cell that result in the expulsion of DNA and antimicrobial proteins. NETosis differs significantly from other forms of cell death like apoptosis and necrosis.
2.2. Key Steps and Molecular Players in NETosis
The formation of NETs involves several critical steps:
-
Activation: Neutrophils are activated by pathogens or inflammatory signals.
-
ROS Generation: NADPH oxidase generates ROS, essential for signaling pathways in NETosis.
-
Nuclear Changes: Neutrophil elastase (NE) and myeloperoxidase (MPO) translocate to the nucleus, contributing to chromatin decondensation.
-
Histone Modification: Peptidylarginine deiminase 4 (PAD4) citrullinates histones, reducing their positive charge and promoting chromatin decondensation.
-
NET Release: The cell membrane ruptures, releasing NETs into the extracellular space.
2.3. Stimuli That Trigger NETosis
NETosis can be triggered by a wide range of stimuli, including:
-
Pathogens: Bacteria, fungi, and viruses.
-
Inflammatory Mediators: Cytokines like TNF-α and interleukins.
-
Phorbol 12-Myristate 13-Acetate (PMA): A potent activator of protein kinase C (PKC).
-
Platelets: Activated platelets interact with neutrophils, inducing NETosis.
2.4. Comparing Suicidal vs. Vital NETosis
NETosis can occur via two primary mechanisms:
-
Suicidal NETosis: Also known as lytic NETosis, involves the death of the neutrophil as it releases NETs. This form typically takes hours and results in the cell’s inability to maintain normal function.
-
Vital NETosis: Allows neutrophils to release NETs while maintaining their cell membrane integrity and continuing to perform functions like phagocytosis. Vital NETosis occurs rapidly and is considered more relevant.
3. The Role of Platelet-Neutrophil Interactions in NETosis
Platelets, traditionally known for their role in hemostasis, also play a significant part in innate immunity by interacting directly with pathogens and promoting NETosis through platelet-neutrophil interactions. These interactions are critical in the context of sepsis, where they can amplify the inflammatory response and contribute to thrombosis.
3.1. Platelets and Their Role in Immunity
Beyond hemostasis, platelets participate in immune responses by interacting with pathogens and releasing various mediators that modulate inflammation. Platelets express Toll-like receptors (TLRs) that recognize pathogen-associated molecular patterns (PAMPs), leading to their activation and subsequent immune responses.
3.2. Mechanisms of Platelet-Neutrophil Interaction
Platelet-neutrophil interactions occur through adhesive and soluble mechanisms:
-
Adhesive Interactions: These involve the binding of platelet adhesion molecules like P-selectin to their neutrophil receptors, such as P-selectin glycoprotein ligand-1 (PSGL-1). Integrins, such as αMβ2 (MAC-1) on neutrophils, also bind to platelet glycoproteins like GPIbα.
-
Soluble Mediators: Activated platelets secrete substances like high mobility group box-1 (HMGB-1) and platelet factor 4 (PF4), which stimulate neutrophils to undergo NETosis.
3.3. How Platelet Activation Influences NETosis
Platelet activation, induced by pathogens or inflammatory signals, leads to the release of chemokines and other mediators that stimulate neutrophils. For instance, platelet-derived CXCL4 and CCL5 activate neutrophils, promoting NETosis. HMGB-1 released by platelets binds to the receptor for advanced glycation endproducts (RAGE) on neutrophils, further inducing NETosis.
3.4. Species-Specific Differences in Platelet-Mediated NETosis
While the general mechanisms of platelet-mediated NETosis are conserved across species, there are notable differences:
- In humans and mice, platelet TLR4 is crucial for LPS-induced NETosis.
- P-selectin/PSGL-1-mediated NETosis appears to be more significant in mice than in humans.
4. The Beneficial Roles of NETs in Sepsis
Despite their potential for harm, NETs have several protective functions during sepsis, primarily related to microbial trapping and direct antimicrobial activity. These benefits highlight the evolutionary role of NETs in host defense.
4.1. Microbial Trapping and Preventing Dissemination
NETs physically trap microorganisms, preventing their spread and dissemination. Studies using high-definition scanning electron microscopy show various pathogens attached to the structural elements of NETs. Mice deficient in PAD4, with reduced ability to produce NETs, are more susceptible to infections, highlighting the protective role of NETs in containing pathogens.
4.2. Direct Antimicrobial Activity of NETs
NETs contain components like histones, cathepsin G, and MPO, which possess bactericidal activities. These components help neutralize and kill entrapped microorganisms. NETs also minimize the pathogenicity of microbes by inactivating their virulence factors.
4.3. Examples of NETs in Action
- In necrotizing fasciitis, NETs enhance the killing of bacteria and ensnare them, hindering their spread.
- In the liver sinusoids of LPS-treated mice, NETs entrap bacteria, potentiating the liver’s ability to clear bacteria during bacteremia.
4.4. Limitations to Antimicrobial Activity
The direct antimicrobial activity of NETs can be limited by factors such as the inactivation of granular proteases by plasma inhibitors. Microbes possessing the ability to rapidly break down DNA are often more virulent, underscoring the importance of NET integrity.
5. The Detrimental Roles of NETs in Sepsis
Excessive NETosis during sepsis can lead to significant harm, including intravascular thrombosis, disseminated intravascular coagulation (DIC), and multiple organ dysfunction. Understanding these detrimental effects is crucial for developing targeted therapies.
5.1. NETs and Thrombosis in Sepsis
Systemic inflammation in sepsis can abnormally activate the coagulation system, leading to excessive intravascular thrombosis. NETs and their components exacerbate DIC by enhancing clot formation, activating platelets, and inhibiting anticoagulant pathways. Septic patients often have elevated levels of circulating cell-free DNA (cfDNA), influencing thrombus formation.
5.2. Mechanisms Linking NETs to Thrombosis
- cfDNA binds to factor XII and high molecular weight kininogen (HMWK), accelerating the activation of the contact pathway of coagulation.
- NETs impair fibrinolysis by inhibiting tissue plasminogen activator.
- Extracellular histones induce platelet activation, aggregation, and thrombin generation via platelet TLR2 and TLR4.
- Neutrophil elastase (NE) and cathepsin G degrade tissue factor pathway inhibitor, further promoting coagulation.
5.3. NETs in Sepsis-Associated Multiple Organ Failure
Aberrant levels of circulating NET components are associated with poor outcomes and multiple organ failure in septic patients. Cell-free DNA (cfDNA) and histones can induce organ damage through various mechanisms:
- cfDNA may contribute to microthrombi formation, leading to microvascular occlusion and tissue hypoxia.
- Histones function as damage-associated molecular patterns, promoting proinflammatory cytokine release, inducing apoptosis, and inciting direct cytotoxicity.
5.4. The Role of NETs in Acute Respiratory Distress Syndrome (ARDS)
Acute respiratory distress syndrome (ARDS) is characterized by disruption of the alveolar-capillary barrier, leading to protein-rich edema and respiratory failure. NETs in bronchoalveolar lavage fluid from septic patients with ARDS indicate that transmigrated neutrophils undergo NETosis, exacerbating lung injury.
6. Potential Therapeutic Targets for NETs in Sepsis
Given the dual role of NETs in sepsis, therapeutic interventions targeting NET production or individual NET components present novel treatment strategies. Clinical assessment of NETs may serve as a valuable biomarker for the early diagnosis of sepsis.
6.1. Targeting Cell-Free DNA (cfDNA)
Elevated levels of cfDNA released from NETosing neutrophils have detrimental effects by activating the coagulation system and inflammation. Studies show that increasing cfDNA clearance using exogenous DNase can improve survival and attenuate organ injury in septic mouse models.
6.2. Inhibiting Peptidylarginine Deiminase 4 (PAD4)
PAD4 is essential for NETosis, making it a suitable therapeutic target. PAD4 inhibitors, such as BB-Cl-amidine, protect against NET-mediated vascular damage and kidney injury in lupus models. Sepsis models utilizing PAD4 knockout mice demonstrate improved survival and decreased organ dysfunction without exacerbating bacteremia.
6.3. Neutralizing Citrullinated Histone H3 (citH3)
Citrullinated histone H3 (citH3) is a potential molecular target due to its proinflammatory, cytotoxic, and prothrombotic properties. Neutralizing circulating citH3 in septic mouse models significantly improves survival. Non-anticoagulant heparin, which binds to extracellular histones, reduces histone-mediated cytotoxicity and improves survival in septic mice.
6.4. Antiplatelet Therapy
Platelet activation and platelet-neutrophil interaction are crucial for NETosis, making antiplatelet therapy a potential strategy. Studies show that inhibiting thromboxane A2 generation with acetylsalicylic acid (aspirin) decreases intravascular NET formation and lung injury. Inhibition of the platelet ADP receptor, P2Y12, may also attenuate platelet-neutrophil interaction and NETosis.
7. Conclusion: Balancing the NET Effect in Sepsis
NETs represent a conserved mechanism of innate immunity with a complex role in sepsis. While they contribute to microbial control, excessive NETosis can exacerbate inflammation, thrombosis, and organ damage. Therapeutic strategies aimed at modulating NET formation and activity hold promise for improving outcomes in sepsis.
7.1. The Future of NET Research
Further research is needed to fully understand the mechanisms that regulate NETosis and the factors that determine whether NETs will be beneficial or detrimental in specific clinical scenarios. This includes exploring novel therapeutic targets and refining existing strategies to balance the immune response in sepsis.
7.2. Seeking Expert Guidance
Navigating the complexities of sepsis treatment requires expert guidance. For more detailed information and tailored advice, contact healthcare professionals or visit reputable medical websites.
7.3. COMPARE.EDU.VN: Your Resource for Informed Decision-Making
At COMPARE.EDU.VN, we understand the challenges in making informed decisions amidst complex medical conditions. That’s why we provide comprehensive comparisons and analyses to help you understand your options and make the best choices for your health and well-being. For more insights, visit our website or contact us at:
- Address: 333 Comparison Plaza, Choice City, CA 90210, United States
- Whatsapp: +1 (626) 555-9090
- Website: COMPARE.EDU.VN
FAQ: Understanding Neutrophil Extracellular Traps (NETs) in Sepsis
1. What are neutrophil extracellular traps (NETs)?
Neutrophil extracellular traps (NETs) are web-like structures composed of DNA, histones, and antimicrobial proteins released by neutrophils to trap and kill pathogens outside the cell.
2. What is NETosis?
NETosis is the process by which neutrophils release NETs. It is a unique form of cell death involving chromatin decondensation and the expulsion of DNA into the extracellular space.
3. How do NETs help fight infection in sepsis?
NETs trap and immobilize pathogens, preventing their spread. They also contain antimicrobial proteins that directly kill or neutralize bacteria, fungi, and viruses.
4. How can NETs be harmful in sepsis?
Excessive NET formation can lead to thrombosis, inflammation, and organ damage. NETs can activate the coagulation system, promote microthrombi formation, and induce cytotoxicity in various organs.
5. What is the role of platelets in NET formation?
Platelets interact with neutrophils and promote NETosis by releasing chemokines and adhesion molecules. Platelet activation enhances NET formation, contributing to the inflammatory response in sepsis.
6. How is cell-free DNA (cfDNA) related to NETs in sepsis?
Cell-free DNA (cfDNA) is a major component of NETs. Elevated levels of cfDNA in septic patients can activate the coagulation system and contribute to organ dysfunction.
7. Can NETs cause acute respiratory distress syndrome (ARDS)?
Yes, NETs can contribute to ARDS by disrupting the alveolar-capillary barrier in the lungs. NET components can degrade surfactant proteins and increase alveolar epithelial permeability, leading to respiratory failure.
8. What are some potential therapeutic targets for NETs in sepsis?
Potential therapeutic targets include:
- Targeting cell-free DNA (cfDNA) with DNase.
- Inhibiting peptidylarginine deiminase 4 (PAD4).
- Neutralizing citrullinated histone H3 (citH3).
- Using antiplatelet therapy to reduce platelet activation and NET formation.
9. Why is early recognition and intervention important in sepsis?
Early recognition and intervention are crucial for improving outcomes in sepsis because they allow for prompt treatment to control infection, modulate the immune response, and prevent organ damage.
10. Where can I find more information about sepsis and NETs?
For more information, visit medical websites like compare.edu.vn or consult with healthcare professionals for tailored advice.
Disclaimer: This article is intended for informational purposes only and does not provide medical advice. Always consult with a qualified healthcare provider for diagnosis and treatment of medical conditions.