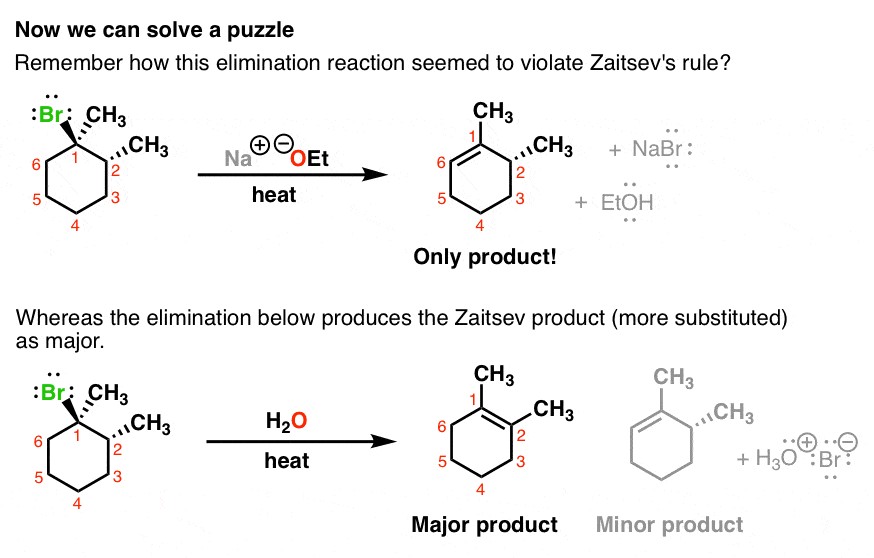
A Chart That Compares Acids And Bases reactions illuminates their fundamental differences, providing clarity for students and professionals alike. At COMPARE.EDU.VN, we offer comprehensive comparisons to help you understand the nuances of chemical reactions. Explore acid-base characteristics, neutralization processes, and titration differences for a deeper understanding.
1. Understanding Acid-Base Chemistry
Acids and bases are fundamental concepts in chemistry, playing a crucial role in numerous chemical reactions and biological processes. Acids are substances that donate protons (H+) or accept electrons, while bases accept protons or donate electrons. These definitions, proposed by Bronsted-Lowry and Lewis respectively, are critical for understanding their behavior in various chemical environments.
- Acids: Substances that increase the concentration of hydrogen ions (H+) in aqueous solutions or accept electrons. They have a pH less than 7, taste sour, and can corrode metals.
- Bases: Substances that decrease the concentration of hydrogen ions (H+) in aqueous solutions or donate electrons. They have a pH greater than 7, taste bitter, and feel slippery.
The reaction between an acid and a base is known as neutralization, which results in the formation of salt and water. This process is essential in many industrial applications, environmental chemistry, and biological systems.
1.1. Defining Acids and Bases
The definitions of acids and bases have evolved over time, with significant contributions from various scientists. Here’s a brief overview of the key definitions:
- Arrhenius Definition: Acids produce H+ ions in water, while bases produce OH- ions. This definition is limited to aqueous solutions.
- Bronsted-Lowry Definition: Acids are proton (H+) donors, and bases are proton acceptors. This definition is broader than the Arrhenius definition.
- Lewis Definition: Acids are electron-pair acceptors, and bases are electron-pair donors. This is the most comprehensive definition, covering reactions in non-aqueous solutions and those not involving proton transfer.
Understanding these definitions is crucial for identifying acids and bases in different chemical contexts. The Bronsted-Lowry definition is particularly useful because it focuses on proton transfer, which is a common feature of acid-base reactions.
1.2. Properties of Acids
Acids exhibit several characteristic properties, including:
- Sour Taste: Acids typically have a sour taste, although tasting them is not recommended due to potential hazards.
- Corrosive Nature: Acids can corrode metals, dissolving them to form salts and hydrogen gas.
- Litmus Paper Test: Acids turn blue litmus paper red.
- pH Value: Acids have a pH value less than 7, indicating a higher concentration of H+ ions.
- Reaction with Bases: Acids react with bases to form salts and water, neutralizing each other.
1.3. Properties of Bases
Bases also have distinct properties:
- Bitter Taste: Bases usually taste bitter, but tasting them is unsafe.
- Slippery Feel: Bases often feel slippery to the touch due to their reaction with oils on the skin.
- Litmus Paper Test: Bases turn red litmus paper blue.
- pH Value: Bases have a pH value greater than 7, indicating a lower concentration of H+ ions.
- Reaction with Acids: Bases react with acids to form salts and water, neutralizing each other.
1.4 The pH Scale and Acid-Base Strength
The pH scale is a measure of how acidic or basic a solution is. It ranges from 0 to 14, with 7 being neutral. Values below 7 indicate acidity, while values above 7 indicate alkalinity (basicity).
- Strong Acids: These acids completely dissociate in water, releasing a large number of H+ ions. Examples include hydrochloric acid (HCl), sulfuric acid (H2SO4), and nitric acid (HNO3).
- Weak Acids: These acids only partially dissociate in water, releasing fewer H+ ions. Examples include acetic acid (CH3COOH) and carbonic acid (H2CO3).
- Strong Bases: These bases completely dissociate in water, releasing a large number of OH- ions. Examples include sodium hydroxide (NaOH) and potassium hydroxide (KOH).
- Weak Bases: These bases only partially dissociate in water, releasing fewer OH- ions. Examples include ammonia (NH3) and pyridine (C5H5N).
The strength of an acid or base is determined by its ability to dissociate in water. Strong acids and bases are highly corrosive and reactive, while weak acids and bases are less so.
2. Key Differences Between Acids and Bases
Acids and bases differ in several key aspects, including their chemical properties, pH values, and reactions with other substances. Here’s a detailed comparison:
| Feature | Acid | Base |
|---|---|---|
| Definition | Proton donor or electron-pair acceptor | Proton acceptor or electron-pair donor |
| Taste | Sour | Bitter |
| Feel | Can be corrosive | Slippery |
| Litmus Paper | Turns blue litmus red | Turns red litmus blue |
| pH Value | Less than 7 | Greater than 7 |
| Neutralization | Reacts with bases to form salts and water | Reacts with acids to form salts and water |
| Examples | HCl, H2SO4, CH3COOH | NaOH, KOH, NH3 |
Understanding these differences is essential for predicting how acids and bases will behave in chemical reactions. The pH value is a particularly important indicator of acidity or basicity, allowing for quantitative comparisons.
2.1. Chemical Properties
The chemical properties of acids and bases dictate their reactivity with other substances. Acids typically react with metals to produce hydrogen gas and salts, while bases react with acids to form salts and water.
- Acids:
- React with metals to produce hydrogen gas and salts.
- React with carbonates to produce carbon dioxide, water, and a salt.
- Neutralize bases to form salts and water.
- Bases:
- React with acids to form salts and water.
- React with ammonium salts to release ammonia gas.
- Can saponify esters, breaking them down into alcohols and carboxylate salts.
These reactions are fundamental to many chemical processes, including industrial synthesis and environmental remediation. For example, the reaction of acids with carbonates is used in the production of carbon dioxide for various applications.
2.2. pH Values
The pH value is a quantitative measure of the acidity or basicity of a solution. It is defined as the negative logarithm (base 10) of the hydrogen ion concentration:
pH = -log[H+]
- Acids: pH values range from 0 to just below 7. Stronger acids have lower pH values.
- Neutral Solutions: pH = 7. This indicates a balance between H+ and OH- ions, as seen in pure water.
- Bases: pH values range from just above 7 to 14. Stronger bases have higher pH values.
The pH scale is logarithmic, meaning that each unit change in pH represents a tenfold change in hydrogen ion concentration. For example, a solution with a pH of 3 has ten times more H+ ions than a solution with a pH of 4.
2.3. Neutralization Reactions
Neutralization is the reaction between an acid and a base, resulting in the formation of salt and water. This process is exothermic, meaning it releases heat. The general equation for a neutralization reaction is:
Acid + Base → Salt + Water
- Example:
- Hydrochloric acid (HCl) reacts with sodium hydroxide (NaOH) to form sodium chloride (NaCl) and water (H2O):
HCl + NaOH → NaCl + H2O
Neutralization reactions are essential in many applications, including:
- Titration: Determining the concentration of an acid or base in a solution.
- Environmental Chemistry: Neutralizing acidic or alkaline pollutants.
- Biological Systems: Maintaining pH balance in living organisms.
3. Factors Affecting Acid-Base Strength
Several factors influence the strength of acids and bases, including molecular structure, electronegativity, and inductive effects. Understanding these factors is crucial for predicting the acidity or basicity of a compound.
3.1. Molecular Structure
The molecular structure of an acid or base plays a significant role in determining its strength. For acids, the stability of the conjugate base is a key factor. The more stable the conjugate base, the stronger the acid.
- Bond Strength: Weaker bonds between hydrogen and the rest of the molecule make it easier to release H+ ions, increasing acidity.
- Polarity: More polar bonds make it easier to remove H+ ions, increasing acidity.
- Resonance: Resonance stabilization of the conjugate base increases acidity by spreading out the negative charge.
For bases, the availability of electrons to accept protons is crucial. Factors that increase electron density on the base molecule enhance its basicity.
3.2. Electronegativity
Electronegativity is the ability of an atom to attract electrons in a chemical bond. In acids, if the atom bonded to hydrogen is highly electronegative, it will pull electron density away from the hydrogen, making it easier to release as H+ ions.
- Example:
- Hydrochloric acid (HCl) is a stronger acid than hydrogen sulfide (H2S) because chlorine is more electronegative than sulfur.
In bases, higher electronegativity on the atom accepting the proton decreases basicity, as it holds onto electrons more tightly.
3.3. Inductive Effects
Inductive effects refer to the transmission of electron density through sigma bonds. Electron-withdrawing groups (EWG) pull electron density away from the acidic proton, stabilizing the conjugate base and increasing acidity.
- Example:
- Trifluoroacetic acid (CF3COOH) is a stronger acid than acetic acid (CH3COOH) because the three fluorine atoms are highly electronegative and withdraw electron density, stabilizing the conjugate base.
Electron-donating groups (EDG) increase electron density on the acidic proton, destabilizing the conjugate base and decreasing acidity.
4. Common Examples of Acids and Bases
Acids and bases are prevalent in everyday life and industrial applications. Here are some common examples:
| Substance | Type | Use |
|---|---|---|
| Hydrochloric Acid (HCl) | Strong Acid | Industrial cleaning, pH control, digestion in the stomach |
| Sulfuric Acid (H2SO4) | Strong Acid | Production of fertilizers, detergents, chemical synthesis |
| Acetic Acid (CH3COOH) | Weak Acid | Vinegar, food preservative, chemical synthesis |
| Citric Acid (C6H8O7) | Weak Acid | Food additive, flavoring agent, cleaning agent |
| Sodium Hydroxide (NaOH) | Strong Base | Soap production, drain cleaner, paper manufacturing |
| Potassium Hydroxide (KOH) | Strong Base | Soap production, electrolyte in alkaline batteries |
| Ammonia (NH3) | Weak Base | Fertilizer, cleaning agent, refrigerant |
| Calcium Hydroxide (Ca(OH)2) | Weak Base | Lime, used in construction, water treatment |
These examples illustrate the diverse roles of acids and bases in various fields. From household cleaning to industrial manufacturing, acids and bases are indispensable chemical compounds.
4.1. Strong Acids
Strong acids completely dissociate in water, releasing a large number of H+ ions. They are highly corrosive and reactive.
- Hydrochloric Acid (HCl): Used in industrial cleaning, pH control, and digestion in the stomach.
- Sulfuric Acid (H2SO4): Used in the production of fertilizers, detergents, and chemical synthesis.
- Nitric Acid (HNO3): Used in the production of fertilizers, explosives, and chemical synthesis.
- Perchloric Acid (HClO4): Used as a strong oxidizing agent and in chemical analysis.
- Hydrobromic Acid (HBr): Used in chemical synthesis and as a catalyst.
- Hydroiodic Acid (HI): Used in chemical synthesis and as a reducing agent.
4.2. Weak Acids
Weak acids only partially dissociate in water, releasing fewer H+ ions. They are less corrosive than strong acids and are often used in food and pharmaceuticals.
- Acetic Acid (CH3COOH): Found in vinegar, used as a food preservative, and in chemical synthesis.
- Citric Acid (C6H8O7): Used as a food additive, flavoring agent, and cleaning agent.
- Carbonic Acid (H2CO3): Formed when carbon dioxide dissolves in water, important in buffering blood pH.
- Phosphoric Acid (H3PO4): Used in fertilizers, detergents, and food additives.
- Formic Acid (HCOOH): Found in ant stings, used in leather tanning and textile dyeing.
- Lactic Acid (C3H6O3): Produced during exercise, used in food preservation and pharmaceuticals.
4.3. Strong Bases
Strong bases completely dissociate in water, releasing a large number of OH- ions. They are highly corrosive and reactive.
- Sodium Hydroxide (NaOH): Used in soap production, drain cleaner, and paper manufacturing.
- Potassium Hydroxide (KOH): Used in soap production and as an electrolyte in alkaline batteries.
- Calcium Hydroxide (Ca(OH)2): Also known as lime, used in construction and water treatment.
- Barium Hydroxide (Ba(OH)2): Used in the synthesis of organic compounds and as a drying agent.
- Strontium Hydroxide (Sr(OH)2): Used in the production of specialty soaps and greases.
4.4. Weak Bases
Weak bases only partially dissociate in water, releasing fewer OH- ions. They are less corrosive than strong bases and are often used in cleaning products and fertilizers.
- Ammonia (NH3): Used in fertilizers, cleaning agents, and as a refrigerant.
- Pyridine (C5H5N): Used as a solvent and in the synthesis of pharmaceuticals and agrochemicals.
- Aniline (C6H5NH2): Used in the production of dyes, plastics, and pharmaceuticals.
- Trimethylamine ((CH3)3N): Found in fish and decaying organic matter, used in chemical synthesis.
5. Applications of Acids and Bases
Acids and bases have numerous applications in various fields, including industrial chemistry, environmental science, and biology.
5.1. Industrial Chemistry
In industrial chemistry, acids and bases are used in a wide range of processes, including:
- Production of Fertilizers: Sulfuric acid and nitric acid are used to produce fertilizers such as ammonium sulfate and ammonium nitrate.
- Synthesis of Polymers: Acids and bases are used as catalysts in the polymerization of monomers to form polymers.
- Petroleum Refining: Sulfuric acid is used to remove impurities from crude oil during refining.
- Metal Processing: Acids are used to etch metals and prepare them for electroplating.
- Production of Detergents: Sulfuric acid and sodium hydroxide are used in the production of detergents.
5.2. Environmental Science
Acids and bases play a crucial role in environmental science, particularly in:
- Water Treatment: Calcium hydroxide (lime) is used to neutralize acidic water and remove impurities.
- Soil Remediation: Acids and bases are used to adjust the pH of soil and improve its fertility.
- Air Pollution Control: Bases are used to scrub acidic gases from industrial emissions.
- Acid Rain Mitigation: Lime is used to neutralize acidic lakes and streams affected by acid rain.
5.3. Biological Systems
Acids and bases are essential for maintaining the proper functioning of biological systems:
- pH Regulation: Buffers, which are mixtures of weak acids and bases, maintain the pH of biological fluids within a narrow range.
- Digestion: Hydrochloric acid in the stomach aids in the digestion of food.
- Enzyme Activity: Enzymes, which catalyze biochemical reactions, are highly sensitive to pH changes.
- Photosynthesis: Carbon dioxide, an acidic gas, is used by plants during photosynthesis to produce glucose and oxygen.
6. Titration: A Quantitative Analysis Technique
Titration is a quantitative analytical technique used to determine the concentration of an acid or base in a solution. It involves the gradual addition of a solution of known concentration (the titrant) to a solution of unknown concentration (the analyte) until the reaction between them is complete. The point at which the reaction is complete is called the equivalence point.
6.1. Types of Titration
There are several types of titration, including:
- Acid-Base Titration: This involves the reaction between an acid and a base. A common example is the titration of hydrochloric acid (HCl) with sodium hydroxide (NaOH).
- Redox Titration: This involves the reaction between an oxidizing agent and a reducing agent. An example is the titration of iron(II) ions with potassium permanganate.
- Complexometric Titration: This involves the formation of a complex between a metal ion and a ligand. An example is the titration of calcium ions with EDTA.
- Precipitation Titration: This involves the formation of a precipitate. An example is the titration of silver ions with chloride ions.
6.2. Indicators in Titration
Indicators are substances that change color at or near the equivalence point, allowing the endpoint of the titration to be visually detected. The choice of indicator depends on the pH range of the equivalence point.
- Phenolphthalein: Changes color from colorless to pink in the pH range of 8.3-10.0, commonly used in titrations involving strong acids and strong bases.
- Methyl Orange: Changes color from red to yellow in the pH range of 3.1-4.4, suitable for titrations involving strong acids and weak bases.
- Bromothymol Blue: Changes color from yellow to blue in the pH range of 6.0-7.6, useful for titrations near neutral pH.
6.3. Performing a Titration
The procedure for performing a titration typically involves the following steps:
- Preparation: Prepare the titrant solution of known concentration and the analyte solution of unknown concentration.
- Setup: Fill a burette with the titrant solution and accurately measure a known volume of the analyte solution into a flask.
- Addition: Gradually add the titrant to the analyte while stirring, monitoring the pH or using an indicator to detect the endpoint.
- Endpoint Determination: Stop adding the titrant when the indicator changes color or the pH reaches the equivalence point.
- Calculation: Use the volume of titrant added and its concentration to calculate the concentration of the analyte.
7. Acid-Base Reactions in Organic Chemistry
Acid-base reactions are fundamental in organic chemistry, influencing reaction mechanisms, catalysis, and synthesis. Understanding these reactions is crucial for predicting the outcome of organic reactions and designing efficient synthetic routes.
7.1. Bronsted-Lowry Acids and Bases in Organic Chemistry
In organic chemistry, Bronsted-Lowry acids and bases are commonly encountered. Organic acids are typically compounds that can donate a proton (H+), while organic bases can accept a proton.
- Carboxylic Acids: These are organic acids that contain a carboxyl group (-COOH). Examples include acetic acid and benzoic acid.
- Alcohols: These can act as weak acids, donating a proton from the hydroxyl group (-OH).
- Amines: These are organic bases that contain a nitrogen atom with a lone pair of electrons. Examples include methylamine and pyridine.
7.2. Lewis Acids and Bases in Organic Chemistry
Lewis acids and bases are also important in organic chemistry. Lewis acids are electron-pair acceptors, while Lewis bases are electron-pair donors.
- Aluminum Chloride (AlCl3): This is a common Lewis acid used as a catalyst in Friedel-Crafts reactions.
- Boron Trifluoride (BF3): Another Lewis acid commonly used as a catalyst in various organic reactions.
- Ethers: These can act as Lewis bases, donating a lone pair of electrons from the oxygen atom.
- Carbonyl Compounds: These can act as Lewis bases, donating a lone pair of electrons from the oxygen atom of the carbonyl group.
7.3. Acid-Base Catalysis
Acid-base catalysis is a common mechanism in organic reactions, where acids or bases are used to accelerate the reaction rate.
- Acid Catalysis: Acids donate protons to activate reactants, making them more susceptible to nucleophilic attack.
- Base Catalysis: Bases remove protons to generate nucleophiles or stabilize leaving groups.
Examples of acid-catalyzed reactions include esterification and hydrolysis, while base-catalyzed reactions include aldol condensation and saponification.
8. The Role of Buffers in Maintaining pH Balance
Buffers are solutions that resist changes in pH when small amounts of acid or base are added. They are crucial in maintaining stable pH levels in biological systems, chemical processes, and environmental applications.
8.1. Composition of Buffers
Buffers typically consist of a weak acid and its conjugate base or a weak base and its conjugate acid. The weak acid or base neutralizes added base or acid, respectively, while the conjugate base or acid neutralizes added acid or base.
- Acidic Buffers: These consist of a weak acid and its conjugate base. An example is acetic acid (CH3COOH) and sodium acetate (CH3COONa).
- Basic Buffers: These consist of a weak base and its conjugate acid. An example is ammonia (NH3) and ammonium chloride (NH4Cl).
8.2. Mechanism of Buffer Action
When an acid is added to a buffer solution, the conjugate base reacts with the added acid, neutralizing it and preventing a significant decrease in pH. Conversely, when a base is added, the weak acid reacts with the added base, neutralizing it and preventing a significant increase in pH.
8.3. Henderson-Hasselbalch Equation
The Henderson-Hasselbalch equation relates the pH of a buffer solution to the pKa of the weak acid and the ratio of the concentrations of the acid and its conjugate base:
pH = pKa + log([A-]/[HA])
where:
- pH is the pH of the buffer solution
- pKa is the negative logarithm of the acid dissociation constant (Ka)
- [A-] is the concentration of the conjugate base
- [HA] is the concentration of the weak acid
This equation is useful for calculating the pH of a buffer solution and for selecting appropriate buffer systems for specific pH ranges.
9. Advanced Concepts in Acid-Base Chemistry
Beyond the basics, acid-base chemistry involves several advanced concepts that are crucial for a deeper understanding of chemical reactions and processes.
9.1. Superacids and Superbases
Superacids are acids that are more acidic than 100% sulfuric acid, while superbases are bases that are stronger than sodium hydroxide. These compounds have unique properties and applications in specialized chemical reactions.
- Superacids: Examples include fluoroantimonic acid (HF·SbF5) and magic acid (FSO3H·SbF5). They are used as catalysts in organic synthesis and in the protonation of unreactive compounds.
- Superbases: Examples include alkyllithium compounds (e.g., butyllithium) and metal amides (e.g., sodium amide). They are used in organic synthesis for deprotonation reactions and in the formation of carbanions.
9.2. Amphoteric Compounds
Amphoteric compounds are substances that can act as both acids and bases, depending on the reaction conditions. Amino acids are a prime example, containing both an acidic carboxyl group (-COOH) and a basic amino group (-NH2).
- Amino Acids: In acidic solutions, the amino group can accept a proton, acting as a base. In basic solutions, the carboxyl group can donate a proton, acting as an acid.
- Water: Water can act as both an acid and a base, donating or accepting protons in various reactions.
9.3. Acid-Base Indicators and Color Change Mechanisms
Acid-base indicators are substances that change color depending on the pH of the solution. The color change is due to the protonation or deprotonation of the indicator molecule, which alters its electronic structure and light absorption properties.
- Phenolphthalein: This indicator is colorless in acidic solutions and pink in basic solutions. The color change is due to the opening of a lactone ring upon deprotonation.
- Methyl Orange: This indicator is red in acidic solutions and yellow in basic solutions. The color change is due to the protonation or deprotonation of a nitrogen atom in the molecule.
10. Practical Guide to Identifying Acids and Bases
Identifying acids and bases in various contexts can be straightforward with a practical approach that combines theoretical knowledge and experimental techniques.
10.1. Visual Inspection and Physical Properties
- Taste: While not recommended due to safety concerns, acids generally have a sour taste, and bases have a bitter taste.
- Touch: Acids can be corrosive, and bases often feel slippery to the touch.
- Odor: Some acids, like acetic acid (vinegar), have a pungent odor. Ammonia, a common base, has a sharp, irritating smell.
10.2. Litmus Paper and pH Indicators
- Litmus Paper: Acids turn blue litmus paper red, while bases turn red litmus paper blue.
- pH Indicators: Use a universal pH indicator solution or paper to get an approximate pH value. This can help determine if a substance is acidic (pH < 7), neutral (pH = 7), or basic (pH > 7).
10.3. Chemical Reactions
- Reaction with Metals: Acids react with many metals to produce hydrogen gas and a metal salt. For example, hydrochloric acid reacts with zinc to produce zinc chloride and hydrogen gas.
- Reaction with Carbonates: Acids react with carbonates to produce carbon dioxide gas, water, and a salt. For example, hydrochloric acid reacts with calcium carbonate (limestone) to produce carbon dioxide, water, and calcium chloride.
- Neutralization Reactions: Acids and bases neutralize each other to form water and a salt. This reaction can be detected by monitoring the pH change.
10.4. Common Examples
- Household Acids: Vinegar (acetic acid), lemon juice (citric acid), and muriatic acid (hydrochloric acid).
- Household Bases: Baking soda (sodium bicarbonate), ammonia, and drain cleaner (sodium hydroxide).
FAQ: Acids and Bases
Q1: What is the difference between a strong acid and a weak acid?
A: Strong acids completely dissociate in water, releasing a large number of H+ ions, while weak acids only partially dissociate.
Q2: What is the pH scale, and how is it used to measure acidity and basicity?
A: The pH scale ranges from 0 to 14, with 7 being neutral. Values below 7 indicate acidity, and values above 7 indicate basicity.
Q3: What is a neutralization reaction?
A: A neutralization reaction is the reaction between an acid and a base, resulting in the formation of salt and water.
Q4: What are some common examples of acids and bases in everyday life?
A: Common examples include hydrochloric acid in the stomach, sodium hydroxide in drain cleaner, and acetic acid in vinegar.
Q5: How do molecular structure and electronegativity affect acid-base strength?
A: Molecular structure and electronegativity influence the stability of the conjugate base or the availability of electrons, affecting the acid or base strength.
Q6: What is the role of buffers in maintaining pH balance?
A: Buffers resist changes in pH by neutralizing small amounts of added acid or base, maintaining a stable pH level.
Q7: What is titration, and how is it used to determine the concentration of an acid or base?
A: Titration is a quantitative analytical technique used to determine the concentration of an acid or base by gradually adding a solution of known concentration to a solution of unknown concentration until the reaction is complete.
Q8: Can a substance be both an acid and a base?
A: Yes, amphoteric compounds can act as both acids and bases, depending on the reaction conditions.
Q9: How do acid-base reactions contribute to organic chemistry?
A: Acid-base reactions influence reaction mechanisms, catalysis, and synthesis in organic chemistry by donating or accepting protons and affecting the reactivity of organic molecules.
Q10: What are superacids and superbases, and what are their applications?
A: Superacids are acids more acidic than 100% sulfuric acid, and superbases are bases stronger than sodium hydroxide. They are used in specialized chemical reactions, such as protonating unreactive compounds and forming carbanions.
Navigating the complexities of acid-base chemistry requires a reliable resource that offers detailed comparisons and insightful analysis. At COMPARE.EDU.VN, we provide comprehensive charts and articles to help you understand the nuances of chemical reactions, empowering you to make informed decisions. Whether you’re a student, educator, or professional, our platform is designed to meet your needs.
Ready to explore more comparisons and make informed decisions? Visit COMPARE.EDU.VN today. Our extensive database and user-friendly interface make it easy to find the information you need. Contact us at 333 Comparison Plaza, Choice City, CA 90210, United States, or reach out via WhatsApp at +1 (626) 555-9090. compare.edu.vn – your ultimate resource for comprehensive comparisons.