Are Geminal Carbons Stable Compared To Cis Trans arrangements in organic molecules? COMPARE.EDU.VN delves into the factors influencing alkene stability, focusing on substitution patterns and isomeric forms to provide clarity. Explore the relative stabilities, considering effects such as steric hindrance and electronic interactions, aiding in a deeper comprehension of chemical behavior, structural isomers and thermodynamic stability.
1. Understanding Alkene Stability
Alkene stability is a cornerstone concept in organic chemistry, crucial for predicting reaction outcomes and understanding molecular behavior. This stability is not inherent but rather influenced by various factors. One significant method to quantify and compare alkene stability involves examining the heat of hydrogenation, a thermodynamic measure that reveals the energy differences between different alkene structures. The concepts of geminal, cis, and trans arrangements each play a unique role in determining an alkene’s overall stability, making this comparison essential for students, consumers, and experts alike.
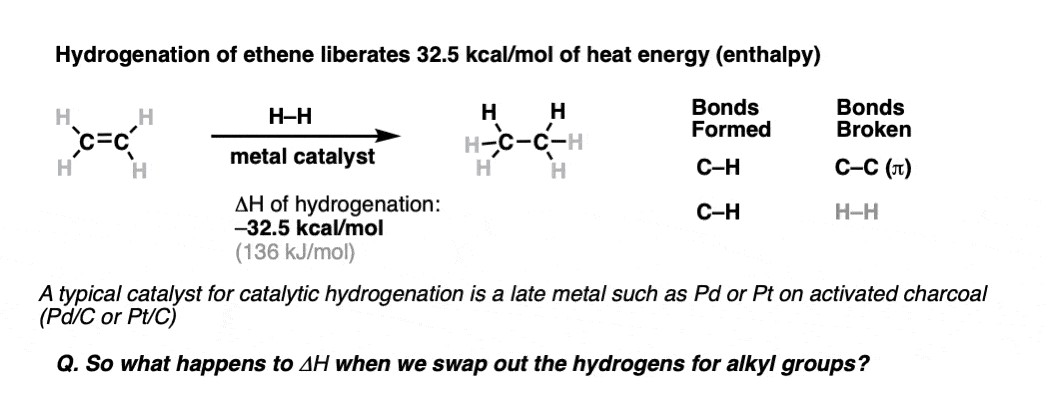
2. Heat of Hydrogenation as an Indicator
The heat of hydrogenation serves as a vital thermodynamic tool in assessing alkene stability. This measurement quantifies the amount of heat released when an alkene is hydrogenated to form an alkane. The magnitude of this heat release is inversely proportional to the stability of the alkene: a lower heat of hydrogenation indicates a more stable alkene, as less energy is released during the saturation process. This is because stable alkenes are already in a lower energy state, requiring less energy input (in the form of hydrogenation) to convert them to their saturated alkane counterparts.
2.1. Factors Influencing Heat of Hydrogenation
Several structural factors contribute to the heat of hydrogenation, including the degree of substitution, steric hindrance, and electronic effects. These factors affect the inherent energy state of the alkene, which is reflected in the amount of heat released upon hydrogenation.
3. The Impact of Alkyl Substitution
The degree of alkyl substitution is a critical determinant of alkene stability. Alkenes with a higher degree of substitution—meaning more alkyl groups attached to the carbon-carbon double bond—are generally more stable. This phenomenon is attributed to hyperconjugation, where the electrons in the adjacent sigma bonds (C-H or C-C) interact with the pi system of the double bond, effectively delocalizing electron density and stabilizing the molecule.
3.1. Stability Order: Tetra- > Tri- > Di- > Mono-Substituted
The stability of alkenes generally follows this order: tetra-substituted > tri-substituted > di-substituted > mono-substituted. Tetra-substituted alkenes, having the maximum number of alkyl substituents, exhibit the greatest stability due to the enhanced hyperconjugation. Mono-substituted alkenes, with only one alkyl substituent, are the least stable.
4. Geminal Carbons and Their Role
Geminal carbons refer to two substituents that are attached to the same carbon atom in a molecule. In the context of alkenes, geminal substitution involves two alkyl groups bonded to one carbon of the double bond, while the other carbon may or may not have additional substituents.
4.1. Geminal vs. Other Disubstituted Alkenes
Compared to other disubstituted alkenes, geminal arrangements have distinct stability implications. The proximity of the two substituents can lead to increased steric hindrance, which may destabilize the molecule compared to trans-disubstituted alkenes where the substituents are on opposite sides of the double bond, minimizing steric interactions. However, the electronic effects of alkyl groups on a geminally substituted carbon can still contribute to stability through hyperconjugation.
5. Cis-Trans Isomerism and Stability
Cis-trans isomerism (also known as geometric isomerism) arises in alkenes when substituents are arranged differently around the double bond. Cis isomers have substituents on the same side of the double bond, while trans isomers have them on opposite sides.
5.1. Trans Isomers: Generally More Stable
Trans isomers are generally more stable than cis isomers due to reduced steric hindrance. In cis isomers, the substituents on the same side of the double bond experience steric repulsion, which raises the energy of the molecule and decreases its stability. This effect is particularly pronounced with bulky substituents.
6. Comparing Geminal, Cis, and Trans Arrangements
When comparing geminal, cis, and trans arrangements, it’s essential to consider both steric and electronic factors. Trans-disubstituted alkenes often represent the most stable configuration due to minimal steric hindrance. Geminal disubstitution can offer stability through hyperconjugation but may be less stable than trans due to steric strain. Cis-disubstituted alkenes are generally the least stable due to significant steric interactions.
6.1. Factors to Consider in Stability Analysis
- Steric Hindrance: The spatial arrangement of substituents and their bulkiness play a crucial role in determining stability.
- Hyperconjugation: The electron-donating effects of alkyl groups through sigma-pi orbital interactions stabilize the alkene.
- Electronic Effects: Inductive effects and other electronic interactions contribute to the overall electron density and stability of the molecule.
7. Case Studies and Examples
To illustrate these principles, consider specific examples of alkenes with different substitution patterns.
7.1. Example 1: 2-Methylpropene (Geminal)
2-Methylpropene, a geminally disubstituted alkene, has two methyl groups attached to one carbon of the double bond. While the methyl groups provide some stabilization through hyperconjugation, their proximity introduces steric strain compared to trans-2-butene.
7.2. Example 2: cis– and trans-2-Butene
Cis-2-butene and trans-2-butene are geometric isomers that provide a clear comparison of cis and trans arrangements. Trans-2-butene is more stable due to the methyl groups being on opposite sides of the double bond, reducing steric hindrance.
7.3. Example 3: 2,3-Dimethyl-2-Butene (Tetrasubstituted)
2,3-Dimethyl-2-butene, a tetrasubstituted alkene, showcases the stabilizing effect of multiple alkyl substituents. This compound is highly stable due to extensive hyperconjugation.
8. Conjugation: An Additional Stabilizing Factor
Conjugation, the presence of alternating single and double bonds in a molecule, introduces additional stability to alkenes. This phenomenon results from the delocalization of pi electrons across the conjugated system, lowering the overall energy of the molecule.
8.1. Resonance Energy in Conjugated Systems
The extra stability conferred by conjugation is quantified as resonance energy, which is the difference between the experimental heat of hydrogenation of a conjugated alkene and the heat of hydrogenation calculated for a non-conjugated analogue.
9. Practical Implications
Understanding the stability of alkenes has numerous practical applications in chemistry and related fields.
9.1. Predicting Reaction Outcomes
The relative stability of alkenes can predict the outcome of reactions such as elimination reactions, where the more stable alkene product is often favored (Zaitsev’s rule).
9.2. Polymer Chemistry
In polymer chemistry, the stability of alkene monomers influences the polymerization process and the properties of the resulting polymers.
9.3. Material Science
In material science, understanding alkene stability is crucial for designing and synthesizing stable and durable materials.
10. Advanced Considerations
For advanced learners, it’s important to consider the more nuanced aspects of alkene stability, such as the effects of ring strain and specific electronic interactions.
10.1. Ring Strain in Cyclic Alkenes
Cyclic alkenes can experience ring strain, particularly in small rings, which affects their stability. Trans-cycloalkenes, for example, are highly strained unless the ring is large enough to accommodate the trans double bond.
10.2. Electronic Interactions and Inductive Effects
Beyond hyperconjugation, inductive effects and other electronic interactions can influence alkene stability. Electron-withdrawing groups can destabilize alkenes, while electron-donating groups can enhance their stability.
11. Common Misconceptions
Addressing common misconceptions is essential for a thorough understanding of alkene stability.
11.1. Misconception: All Disubstituted Alkenes Are Equally Stable
It is a common misconception that all disubstituted alkenes are equally stable. In reality, trans-disubstituted alkenes are generally more stable than cis-disubstituted and geminal alkenes due to reduced steric hindrance.
11.2. Misconception: Substitution Always Increases Stability
While increasing substitution generally increases stability, this is not always the case. Bulky substituents can introduce steric strain, which can counteract the stabilizing effects of hyperconjugation.
12. Alkene Stability and Zaitsev’s Rule
Zaitsev’s rule is a crucial concept related to alkene stability. It states that in an elimination reaction, the major product is the more substituted alkene. This rule is based on the principle that more substituted alkenes are thermodynamically more stable.
12.1. Application of Zaitsev’s Rule
Zaitsev’s rule is widely applied in predicting the products of elimination reactions, particularly in organic synthesis. Understanding alkene stability helps predict the major product in these reactions.
13. Spectroscopic Analysis of Alkenes
Spectroscopic techniques such as NMR and IR spectroscopy can provide valuable information about the structure and stability of alkenes.
13.1. NMR Spectroscopy
NMR spectroscopy can distinguish between different types of alkenes based on the chemical shifts of the vinylic protons and carbons.
13.2. IR Spectroscopy
IR spectroscopy can identify the presence of a carbon-carbon double bond based on the characteristic stretching frequency around 1650 cm-1.
14. Alkene Stability in Biological Systems
Alkenes play a crucial role in various biological systems. Understanding their stability is essential for comprehending biochemical processes.
14.1. Fatty Acids
Unsaturated fatty acids contain carbon-carbon double bonds. The cis or trans configuration of these double bonds affects the physical properties and biological functions of the fatty acids.
14.2. Terpenes and Steroids
Terpenes and steroids, which are important biological molecules, often contain alkene functionalities. The stability and reactivity of these alkenes influence the biological activity of these compounds.
15. Modern Research and Developments
Modern research continues to explore novel aspects of alkene stability and reactivity.
15.1. Catalysis
Catalysis plays a crucial role in many reactions involving alkenes. Researchers are developing new catalysts that can selectively react with alkenes based on their stability and substitution patterns.
15.2. Green Chemistry
Green chemistry initiatives aim to develop more sustainable and environmentally friendly methods for synthesizing and using alkenes.
16. Expert Insights on COMPARE.EDU.VN
At COMPARE.EDU.VN, we provide in-depth analyses of chemical concepts, including alkene stability, to empower students, consumers, and experts with comprehensive and reliable information. Our comparisons are designed to help you make informed decisions based on the latest scientific knowledge.
16.1. Utilizing COMPARE.EDU.VN for Chemical Comparisons
COMPARE.EDU.VN offers detailed comparisons of different chemical compounds and their properties. Our platform allows you to explore the nuances of alkene stability and compare the characteristics of various isomeric forms.
17. Real-World Applications of Alkene Stability
Understanding alkene stability is not just an academic exercise; it has real-world applications in various industries.
17.1. Petrochemical Industry
In the petrochemical industry, alkene stability is crucial for optimizing the production of fuels and chemicals.
17.2. Pharmaceutical Industry
In the pharmaceutical industry, alkene stability influences the design and synthesis of drugs and other therapeutic agents.
18. Impact of Steric Hindrance on Alkene Stability
Steric hindrance is a crucial factor in determining the stability of alkenes. It refers to the spatial arrangement of atoms within a molecule that can cause repulsive interactions due to the occupancy of the same space. In alkenes, particularly those with bulky substituents, steric hindrance can significantly destabilize the molecule.
18.1. Bulky Substituents and Destabilization
Bulky substituents attached to the carbon atoms of the double bond can lead to steric strain, where the electron clouds of the substituents repel each other. This repulsion raises the energy of the molecule, making it less stable. For example, in cis-substituted alkenes, the substituents are on the same side of the double bond, leading to significant steric hindrance if the groups are large.
18.2. Comparison with Trans Isomers
Trans isomers, where the substituents are on opposite sides of the double bond, generally experience less steric hindrance. This reduced steric strain contributes to the higher stability of trans alkenes compared to their cis counterparts. The difference in stability can be significant, particularly with large, bulky substituents.
19. Electronic Effects on Alkene Stability
Electronic effects, such as inductive effects and resonance, also play a significant role in determining alkene stability. These effects influence the distribution of electron density within the molecule, affecting its overall stability.
19.1. Inductive Effects
Inductive effects refer to the polarization of sigma bonds due to the electronegativity differences between atoms. Alkyl groups are electron-donating, meaning they can donate electron density to the double bond, stabilizing it. The more alkyl groups attached to the alkene, the greater the electron donation, and the more stable the alkene becomes.
19.2. Resonance and Conjugation
Resonance occurs when electrons can be delocalized over multiple atoms, such as in conjugated systems. Conjugated alkenes, with alternating single and double bonds, are more stable due to the delocalization of pi electrons across the system. This delocalization lowers the overall energy of the molecule, increasing its stability.
20. Quantifying Alkene Stability: Thermodynamic Data
Thermodynamic data, such as heats of hydrogenation and heats of formation, provide quantitative measures of alkene stability. These values can be used to compare the relative stabilities of different alkenes and to understand the effects of substitution, steric hindrance, and electronic effects.
20.1. Heats of Hydrogenation
The heat of hydrogenation is the amount of heat released when an alkene is hydrogenated to form an alkane. A lower (more negative) heat of hydrogenation indicates a more stable alkene, as it requires less energy to convert it to the alkane.
20.2. Heats of Formation
The heat of formation is the change in enthalpy when a compound is formed from its constituent elements in their standard states. More stable compounds have lower (more negative) heats of formation.
21. How to Predict Alkene Stability: A Step-by-Step Guide
Predicting alkene stability involves considering several factors. Here is a step-by-step guide to help you make accurate predictions:
21.1. Assess the Degree of Substitution
First, determine the degree of substitution of the alkene. More substituted alkenes are generally more stable due to hyperconjugation.
21.2. Evaluate Steric Hindrance
Next, evaluate the amount of steric hindrance in the molecule. Cis isomers and alkenes with bulky substituents may be less stable due to steric strain.
21.3. Consider Electronic Effects
Finally, consider the electronic effects, such as inductive effects and resonance. Alkyl groups can stabilize the alkene through electron donation, while conjugation can enhance stability through electron delocalization.
22. Common Reactions Involving Alkenes and Their Stability
Alkene stability influences the outcome of many chemical reactions. Understanding this influence can help predict the products and mechanisms of these reactions.
22.1. Addition Reactions
Addition reactions involve the addition of atoms or groups of atoms across the double bond of an alkene. The stability of the alkene affects the rate and selectivity of these reactions.
22.2. Elimination Reactions
Elimination reactions involve the removal of atoms or groups of atoms from a molecule, resulting in the formation of an alkene. The stability of the resulting alkene influences which product will be favored, as described by Zaitsev’s rule.
23. Advanced Techniques for Analyzing Alkene Stability
For those interested in delving deeper into the analysis of alkene stability, advanced techniques such as computational chemistry and X-ray crystallography can provide valuable insights.
23.1. Computational Chemistry
Computational chemistry methods, such as density functional theory (DFT), can be used to calculate the energies of different alkene conformations and to predict their relative stabilities.
23.2. X-Ray Crystallography
X-ray crystallography can provide detailed structural information about alkenes, including bond lengths, bond angles, and substituent arrangements. This information can be used to assess the degree of steric hindrance and to understand the factors influencing alkene stability.
24. Alkene Stability and Polymerization
Alkene stability is a crucial consideration in polymerization reactions, where alkenes are used as monomers to form polymers. The stability of the alkene monomer affects the ease and selectivity of the polymerization process.
24.1. Types of Polymerization
Different types of polymerization reactions, such as addition polymerization and condensation polymerization, are used to synthesize polymers from alkenes. The choice of polymerization method depends on the structure and stability of the alkene monomer.
24.2. Polymer Properties
The properties of the resulting polymer, such as its strength, flexibility, and thermal stability, are influenced by the structure and stability of the alkene monomer.
25. FAQs About Alkene Stability
Q1: What is the primary factor that determines alkene stability?
A: The primary factor is the degree of substitution. More substituted alkenes are generally more stable due to hyperconjugation.
Q2: Why are trans alkenes more stable than cis alkenes?
A: Trans alkenes are more stable than cis alkenes due to reduced steric hindrance.
Q3: How does conjugation affect alkene stability?
A: Conjugation increases alkene stability by allowing for the delocalization of pi electrons across the conjugated system.
Q4: What is Zaitsev’s rule?
A: Zaitsev’s rule states that in an elimination reaction, the major product is the more substituted alkene.
Q5: How can heat of hydrogenation be used to determine alkene stability?
A: A lower heat of hydrogenation indicates a more stable alkene.
Q6: What are geminal carbons in the context of alkenes?
A: Geminal carbons refer to two substituents that are attached to the same carbon atom in an alkene.
Q7: How does steric hindrance affect alkene stability?
A: Steric hindrance destabilizes alkenes, especially when bulky substituents are on the same side of the double bond (cis isomers).
Q8: What are the key electronic effects influencing alkene stability?
A: Key electronic effects include inductive effects (electron donation from alkyl groups) and resonance (electron delocalization in conjugated systems).
Q9: Can cyclic alkenes have trans configurations?
A: Yes, but trans-cycloalkenes are highly strained unless the ring is large enough to accommodate the trans double bond.
Q10: Why is understanding alkene stability important in organic chemistry?
A: Understanding alkene stability is crucial for predicting reaction outcomes, designing organic syntheses, and comprehending the properties of organic molecules.
26. Conclusion: Mastering Alkene Stability
Mastering the principles of alkene stability is essential for anyone studying or working in chemistry and related fields. By understanding the effects of substitution, steric hindrance, electronic effects, and conjugation, you can accurately predict the stability of alkenes and their behavior in chemical reactions.
Remember, COMPARE.EDU.VN is here to provide you with detailed comparisons and expert insights to help you navigate the complexities of chemistry. Whether you are a student, consumer, or expert, our platform is designed to empower you with the knowledge you need to make informed decisions.
Are you struggling to compare different chemical compounds or understand their properties? Visit COMPARE.EDU.VN today! Our comprehensive comparisons and expert analyses will help you make informed decisions. Don’t let confusion hold you back. Discover the power of clarity with COMPARE.EDU.VN and unlock your potential for success in chemistry and beyond. Visit us at compare.edu.vn or contact us at 333 Comparison Plaza, Choice City, CA 90210, United States. You can also reach us via Whatsapp at +1 (626) 555-9090. Let us help you make the right choice!